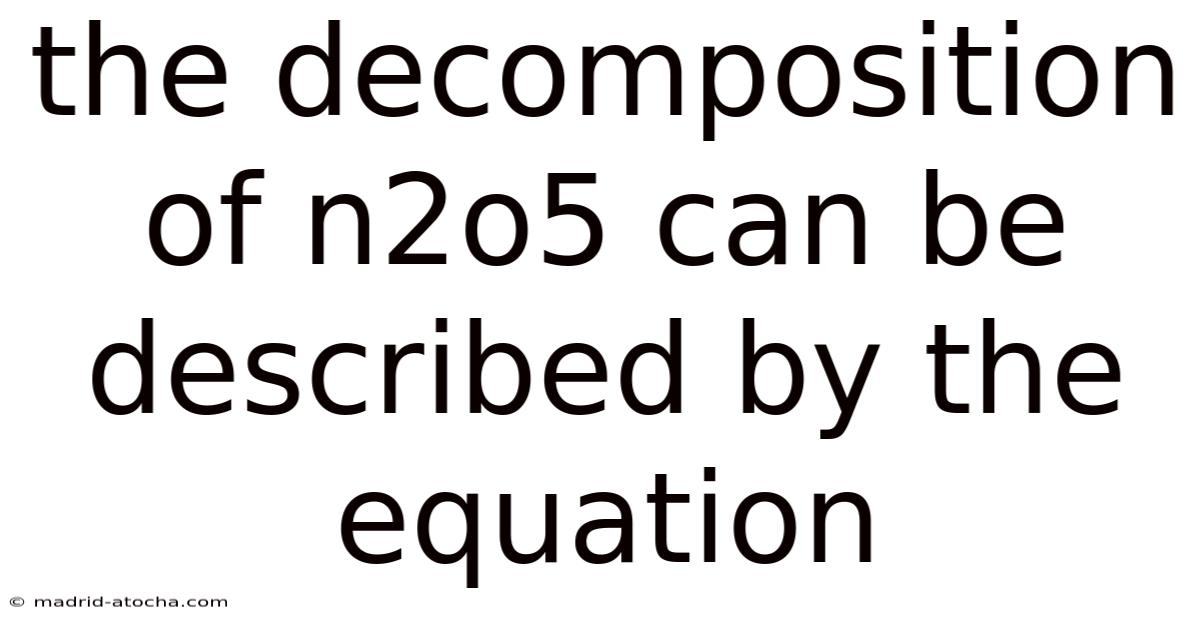The Decomposition of N2O5: A Detailed Look at the Chemical Reaction
The decomposition of nitrogen pentoxide (N2O5) is a fascinating chemical process that has been extensively studied in the field of chemistry. Consider this: this reaction not only serves as an excellent example of a chemical decomposition process but also provides valuable insights into the behavior of chemical compounds under different conditions. In this article, we will break down the details of the decomposition of N2O5, exploring the chemical equation, the reaction mechanism, and the factors that influence this reaction Surprisingly effective..
Introduction to N2O5 Decomposition
Nitrogen pentoxide, with the chemical formula N2O5, is a highly reactive compound that readily decomposes into nitrogen dioxide (NO2) and oxygen (O2). The decomposition of N2O5 is a classic example of a chemical reaction that can be described by a balanced chemical equation. Understanding this reaction is crucial for anyone studying chemistry, as it provides a foundation for understanding more complex chemical processes.
Some disagree here. Fair enough.
The decomposition of N2O5 is a vital topic in the study of chemical kinetics and thermodynamics, as it allows us to explore the rate at which the reaction occurs and the energy changes associated with it. By examining this reaction in detail, we can gain a deeper understanding of the principles that govern chemical reactions and the factors that influence their occurrence.
People argue about this. Here's where I land on it.
The Chemical Equation for N2O5 Decomposition
The balanced chemical equation for the decomposition of N2O5 is as follows:
[ 2N2O5(g) \rightarrow 4NO2(g) + O2(g) ]
This equation shows that two molecules of nitrogen pentoxide decompose to form four molecules of nitrogen dioxide and one molecule of oxygen gas. Because of that, the reaction is exothermic, meaning that it releases energy in the form of heat. This energy release is a result of the breaking of chemical bonds in the reactants and the formation of new bonds in the products.
Reaction Mechanism
The decomposition of N2O5 is a complex reaction that involves multiple steps. The reaction mechanism provides a detailed description of how the reaction proceeds at the molecular level. In the case of N2O5 decomposition, the reaction mechanism involves the following steps:
-
The first step involves the homolytic cleavage of the N2O5 molecule, resulting in the formation of two nitrogen dioxide (NO2) radicals and an oxygen atom. This step is initiated by the breaking of the N-O bonds in N2O5 That's the part that actually makes a difference. No workaround needed..
-
The second step involves the dimerization of the nitrogen dioxide radicals, forming a molecule of nitrogen dioxide dimer (N2O4). This step is driven by the tendency of the radicals to form more stable molecules.
-
The final step involves the further decomposition of the nitrogen dioxide dimer, resulting in the formation of nitrogen dioxide and oxygen gas. This step is driven by the release of energy in the form of heat It's one of those things that adds up..
The reaction mechanism for the decomposition of N2O5 is a classic example of a radical chain reaction, where the initial activation of the reaction leads to the formation of reactive intermediates that drive the reaction forward. Understanding the reaction mechanism is essential for predicting the behavior of the reaction under different conditions and for designing processes that involve the decomposition of N2O5 Most people skip this — try not to..
Factors Affecting N2O5 Decomposition
Several factors can influence the decomposition of N2O5, including temperature, pressure, and the presence of catalysts. The decomposition of N2O5 is an exothermic reaction, meaning that it releases energy in the form of heat. On the flip side, as a result, the reaction rate is highly sensitive to temperature changes. Increasing the temperature of the reaction mixture can accelerate the decomposition of N2O5, while decreasing the temperature can slow down the reaction.
Pressure also is key here in the decomposition of N2O5. Increasing the pressure of the reaction mixture can increase the rate of decomposition, while decreasing the pressure can slow down the reaction. This is because the decomposition of N2O5 is a gas-phase reaction, and the rate of reaction is influenced by the concentration of the reactants and products in the gas phase.
The presence of catalysts can also significantly influence the decomposition of N2O5. On top of that, catalysts are substances that increase the rate of a chemical reaction without being consumed in the reaction. In the case of N2O5 decomposition, catalysts can lower the activation energy of the reaction, making it easier for the reactants to form the products. This can lead to a significant increase in the reaction rate, even at lower temperatures and pressures.
Applications of N2O5 Decomposition
The decomposition of N2O5 has several important applications in the field of chemistry. Day to day, when it comes to applications, in the production of nitrogen dioxide, which is a highly reactive gas with numerous industrial uses is hard to beat. Nitrogen dioxide is a key component in the production of nitric acid, which is used in the production of fertilizers, explosives, and other chemicals Practical, not theoretical..
The decomposition of N2O5 is also important in the study of atmospheric chemistry. Practically speaking, the decomposition of N2O5 can lead to the formation of nitrogen dioxide, which can contribute to the depletion of the ozone layer. Which means nitrogen pentoxide is a highly reactive compound that can be found in the upper atmosphere. Understanding the decomposition of N2O5 is essential for predicting the behavior of the atmosphere and for developing strategies to mitigate the effects of ozone depletion.
Conclusion
The decomposition of nitrogen pentoxide (N2O5) is a fascinating chemical reaction that has numerous applications in the field of chemistry. By understanding the chemical equation, the reaction mechanism, and the factors that influence this reaction, we can gain a deeper understanding of the principles that govern chemical reactions and the factors that influence their occurrence. The decomposition of N2O5 serves as an excellent example of a chemical reaction that can be studied in detail, providing valuable insights into the behavior of chemical compounds under different conditions.
Conclusion
The decomposition of nitrogen pentoxide (N2O5) is a fascinating chemical reaction that has numerous applications in the field of chemistry. Practically speaking, by understanding the chemical equation, the reaction mechanism, and the factors that influence this reaction – including temperature, pressure, and the presence of catalysts – we can gain a deeper understanding of the principles that govern chemical reactions and the factors that influence their occurrence. The decomposition of N2O5 serves as an excellent example of a chemical reaction that can be studied in detail, providing valuable insights into the behavior of chemical compounds under different conditions.
Beyond its role in industrial chemical production and atmospheric science, the study of N2O5 decomposition also contributes to fundamental research in kinetics and reaction mechanisms. It provides a model system for investigating the influence of various parameters on reaction rates and pathways. Adding to this, advancements in understanding this reaction can inform the development of novel catalytic processes and contribute to more efficient and sustainable chemical technologies. Continued research into N2O5 decomposition promises to get to further applications and deepen our comprehension of chemical reactivity in diverse environments Took long enough..
This is the bit that actually matters in practice It's one of those things that adds up..
Thedecomposition of N2O5 also holds relevance in environmental monitoring and pollution control. By optimizing conditions that accelerate or inhibit its breakdown, scientists aim to develop more efficient technologies for mitigating nitrogen-based pollutants, which are key contributors to acid rain and global warming. Researchers are exploring ways to harness the controlled decomposition of N2O5 in catalytic converters or scrubber systems to reduce harmful emissions from vehicles and factories. Now, as industrial activities continue to release nitrogen oxides into the atmosphere, tracking the behavior of compounds like N2O5 becomes critical for assessing air quality and climate impacts. This intersection of chemistry and environmental science underscores the compound’s dual role as both a chemical curiosity and a practical tool for addressing modern ecological challenges.
The official docs gloss over this. That's a mistake.
Conclusion
The decomposition of nitrogen pentoxide (N2O5) exemplifies the complex relationship between chemical theory and real-world application. From its foundational role in industrial chemistry to its implications for atmospheric stability and environmental health, N2O5 decomposition offers a multifaceted lens through which to examine reaction dynamics. Its study not only advances our understanding of kinetic processes and catalytic behavior but also informs strategies to combat climate change and pollution. As research evolves, the ability to manipulate and predict this reaction will remain vital in developing sustainable chemical processes and safeguarding planetary ecosystems. When all is said and done, the exploration of N2O5 decomposition reminds us that even the most seemingly esoteric chemical reactions can have profound, far-reaching consequences for both science and society That's the whole idea..