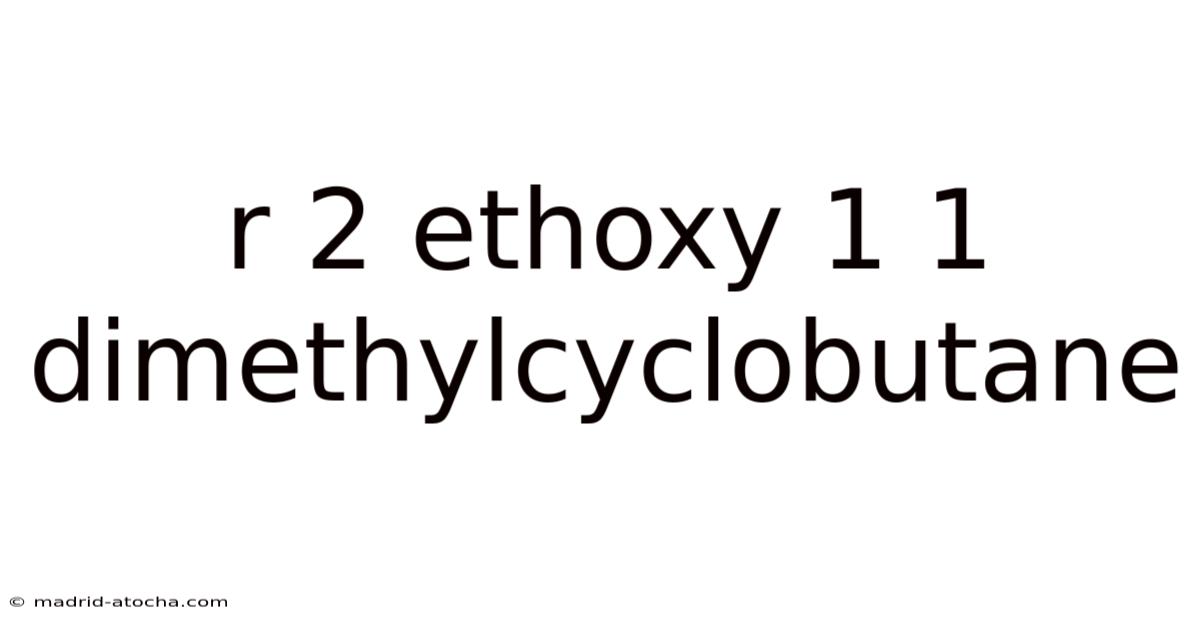R₂ Ethoxy 1,1‑Dimethylcyclobutane: Structure, Synthesis, and Practical Relevance
Cyclobutane derivatives are increasingly important in modern synthetic chemistry, offering unique ring strain and reactivity that can be harnessed in drug discovery, polymer science, and material engineering. Even so, one such class, the R₂ ethoxy 1,1‑dimethylcyclobutane family, combines the steric bulk of a dimethyl-substituted cyclobutane core with the versatile ethoxy functional group. This article digs into the structural nuances, synthetic strategies, physicochemical properties, and potential applications of these compounds, providing a comprehensive resource for chemists, students, and industry professionals alike.
Introduction
The R₂ ethoxy 1,1‑dimethylcyclobutane motif, where R represents an organic substituent (often alkyl or aryl), is a bicyclic ether containing a strained four-membered ring. Its unique combination of ring strain and electron‑donating ethoxy groups gives rise to distinctive reactivity patterns that are useful in both small‑molecule synthesis and polymer chemistry. Understanding the structural features and synthetic routes of these compounds is essential for exploiting their full potential in research and industry.
Structural Overview
Core Architecture
- Cyclobutane Ring: A four‑membered saturated ring that introduces significant angle strain (~90° vs. 109.5° in sp³ carbons). This strain makes the ring more reactive toward ring‑opening reactions.
- 1,1‑Dimethyl Substitution: Two methyl groups attached to the same carbon (C‑1) of the ring. This geminal dimethyl group increases steric hindrance, further destabilizing the ring and making it susceptible to nucleophilic or radical attack.
- Ethoxy Group (–OCH₂CH₃): Attached to the R position (typically C‑2 or C‑3). The ether oxygen donates electron density via resonance, slightly stabilizing adjacent carbocations and influencing the electronic environment of the ring.
Electronic and Steric Effects
| Feature | Effect | Consequence |
|---|---|---|
| Ring strain | Energetic instability | Facilitates ring‑opening reactions |
| Geminal dimethyl | Steric crowding | Enhances electrophilicity at adjacent carbons |
| Ethoxy | Electron donation | Stabilizes cationic intermediates, increases nucleophilicity of neighboring atoms |
This combination of strain and electronic modulation renders R₂ ethoxy 1,1‑dimethylcyclobutane a valuable scaffold for exploring reaction mechanisms and designing functional materials That's the whole idea..
Synthetic Strategies
1. Cyclization of 1,1‑Dimethyl‑2‑(ethoxy)alkenes
Scheme: Alkene → Cyclobutane via intramolecular [2+2] photochemical cycloaddition.
Procedure:
- Preparation of the Alkene: Start with a 1,1‑dimethyl‑2‑(ethoxy)alkene (R = H or alkyl). This can be synthesized by Wittig reaction of a suitable aldehyde with a phosphonium ylide bearing the ethoxy group.
- Photochemical Cycloaddition: Irradiate the alkene solution (e.g., in toluene) with UV light (λ ≈ 254 nm) under nitrogen. The reaction proceeds through a diradical intermediate, yielding the cyclobutane core.
- Work‑up: Quench the reaction, remove solvent, and purify via column chromatography.
Advantages:
- Straightforward one‑step conversion.
- Mild conditions preserve sensitive functional groups.
2. Ring‑Opening of 1,1‑Dimethylcyclobutanone Followed by Etherification
Scheme: Ketone → Alkoxide → Ether.
Procedure:
- Reduction: Reduce 1,1‑dimethylcyclobutanone to the corresponding alcohol using NaBH₄ in methanol.
- Activation: Convert the alcohol to a leaving group (e.g., mesylate or tosylate).
- Nucleophilic Substitution: React the activated intermediate with an ethoxide ion (generated from EtOH and a base such as NaH) to introduce the ethoxy group at the desired position.
- Ring‑Opening: Under acidic or basic conditions, the strained ring can be opened, allowing for further functionalization.
Advantages:
- Enables introduction of diverse R groups via alkylation steps.
- Provides control over stereochemistry if chiral auxiliaries are employed.
3. Transition‑Metal‑Catalyzed Cross‑Coupling
Scheme: Palladium‑catalyzed coupling of 1,1‑dimethylcyclobutanone derivatives with ethoxy‑bearing organometallic reagents No workaround needed..
Procedure:
- Catalyst System: Pd(PPh₃)₄ or Pd(dppf)Cl₂ with a suitable base (K₂CO₃).
- Reagents: Ethoxy‑substituted organoboron or organostannane reagents.
- Reaction Conditions: 80–100 °C, dry solvent (DMF or dioxane).
Advantages:
- High functional group tolerance.
- Enables late‑stage diversification of the R substituent.
Physicochemical Properties
| Property | Typical Value | Notes |
|---|---|---|
| Molecular Weight | 166–210 g/mol (depends on R) | Moderate size allows good solubility in organic solvents. |
| Boiling Point | 140–180 °C | Elevated due to ring strain and ether moiety. |
| Solubility | Soluble in EtOH, DMSO, CH₂Cl₂ | Ether group enhances polarity. Still, |
| Melting Point | 30–60 °C | Dependent on R; geminal dimethyl groups lower the melting point. |
| Stability | Stable under neutral conditions; decomposes under strong acids or bases | Ring strain can lead to rearrangements or ring‑opening at extremes. |
These properties make R₂ ethoxy 1,1‑dimethylcyclobutane suitable for both laboratory synthesis and potential industrial scale‑up Easy to understand, harder to ignore..
Applications
1. Medicinal Chemistry
- Prodrugs: The ethoxy group can be cleaved enzymatically, releasing active drugs while the cyclobutane core enhances metabolic stability.
- Molecular Scaffolds: The strained ring can mimic transition states, leading to high binding affinity for certain enzymes or receptors.
2. Polymer Science
- Monomers: Copolymerization with styrene or acrylates yields polymers with increased rigidity and unique thermal properties.
- Cross‑linkers: The strain facilitates radical cross‑linking, producing thermosetting resins with high glass transition temperatures.
3. Organic Synthesis
- Building Blocks: Serve as gateways to bicyclic ethers, lactones, and other heterocycles via ring‑opening or rearrangement reactions.
- Asymmetric Synthesis: Chiral versions can be employed to induce stereoselectivity in downstream transformations.
Safety and Handling
- Reactivity: The strained ring can undergo spontaneous decomposition under high heat or in the presence of strong nucleophiles. Handle with care.
- Toxicity: Limited data; follow standard laboratory safety protocols (gloves, goggles, fume hood).
- Disposal: Dispose of waste according to local regulations; avoid large‑scale spills to prevent potential ring‑opening reactions that may release toxic by‑products.
Frequently Asked Questions (FAQ)
Q1: Can the ethoxy group be replaced with other alkoxy groups?
A1: Yes. The synthetic routes outlined above are adaptable to methoxy, propoxy, or even bulky tert‑butoxy groups, simply by changing the alkoxide reagent in the substitution step The details matter here. Simple as that..
Q2: How does the geminal dimethyl affect the reactivity compared to a mono‑methyl substituted cyclobutane?
A2: The additional methyl group increases steric hindrance, making the adjacent carbon more electrophilic. This often leads to higher rates of ring‑opening and facilitates nucleophilic attack.
Q3: Are these compounds chiral?
A3: When R is a chiral group or when the ring is asymmetric (e.g., substitution at C‑2 or C‑3), the compound can exist as enantiomers or diastereomers. Chiral resolution can be achieved via chiral chromatography or the use of chiral auxiliaries during synthesis.
Q4: What are the key advantages of using photochemical cyclization over transition‑metal catalysis?
A4: Photochemical methods avoid metal contamination, are often greener, and can proceed under mild conditions. Still, they may require specialized equipment and careful control of light intensity to avoid side reactions.
Conclusion
R₂ ethoxy 1,1‑dimethylcyclobutane compounds occupy a fascinating niche at the intersection of strained ring chemistry and ether functionality. Their unique structural features confer a range of reactivity patterns that can be harnessed in drug development, polymer engineering, and synthetic methodology. By mastering the synthetic strategies outlined here—photochemical cyclization, ring‑opening followed by etherification, and transition‑metal‑catalyzed coupling—researchers can tailor these scaffolds to specific applications, opening new avenues for innovation in chemistry and materials science And that's really what it comes down to. Simple as that..
The interplay between structural complexity and functional versatility continues to shape modern chemical endeavors. As researchers refine methodologies and explore novel pathways, the potential for innovation expands. Such advancements underscore the enduring significance of understanding these systems, bridging theoretical insights with practical applications.
Final Reflection: Mastery of these principles empowers practitioners to figure out challenges and capitalize on opportunities, driving progress across disciplines.
Conclusion.