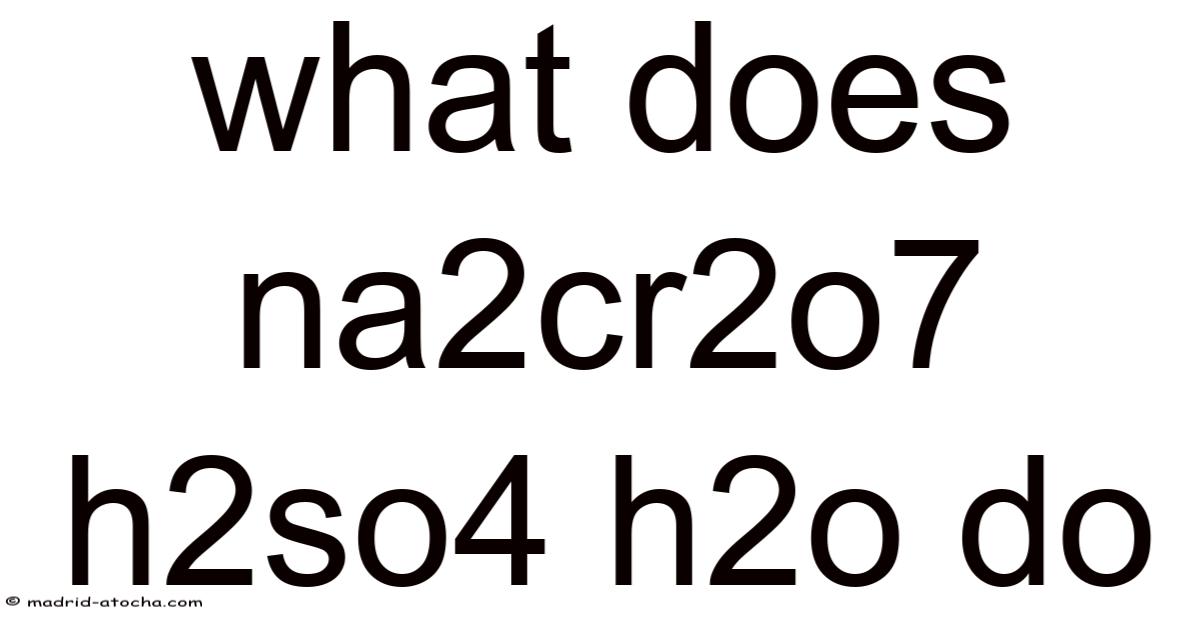What Does Na2Cr2O7 + H2SO4 + H2O Do? A Complete Guide to This Powerful Chemical Combination
The chemical combination of sodium dichromate (Na2Cr2O7), sulfuric acid (H2SO4), and water (H2O) is one of the most important reagent systems in organic chemistry. This powerful mixture serves as a source of chromic acid, a versatile oxidizing agent that matters a lot in numerous chemical transformations. Understanding what this combination does and how it works is essential for students and professionals working in analytical and synthetic chemistry.
Understanding the Chemical Components
Before exploring what happens when these chemicals combine, it is important to understand each component individually and its role in the reaction mixture Still holds up..
Sodium dichromate (Na2Cr2O7) is an inorganic compound that exists as orange-red crystals. This compound contains chromium in its +6 oxidation state, which is responsible for its strong oxidizing properties. Sodium dichromate is highly soluble in water and serves as a convenient source of chromate ions (CrO4²⁻) and dichromate ions (Cr2O7²⁻) in solution.
Sulfuric acid (H2SO4) is a strong mineral acid that acts as both a catalyst and a proton source in this reaction system. Its role extends beyond simple acid catalysis—it helps convert the dichromate ions into the active oxidizing species while also providing the necessary acidic environment for oxidation reactions to proceed efficiently.
Water (H2O) serves as the solvent and is essential for the formation of chromic acid. The presence of water determines whether the reaction produces chromic acid (H2CrO4) or chromium trioxide (CrO3), both of which function as oxidizing agents.
The Chemical Reaction: What Actually Happens
When sodium dichromate reacts with sulfuric acid in the presence of water, a series of important chemical transformations occur. The primary reaction produces chromic acid, a powerful oxidizing agent that is responsible for most of the chemical activity associated with this mixture.
The simplified overall reaction can be represented as:
Na2Cr2O7 + 2H2SO4 → 2CrO3·H2O + 2NaHSO4
In this equation, CrO3·H2O represents chromic acid (H2CrO4), which exists in equilibrium with chromium trioxide and water. The reaction also produces sodium bisulfate (NaHSO4) as a byproduct.
The chemistry becomes more nuanced when considering the equilibrium between different chromium(VI) species in solution:
Cr2O7²⁻ + H2O ⇌ 2CrO4²⁻ + 2H⁺
This equilibrium shows that dichromate ions (Cr2O7²⁻) can convert to chromate ions (CrO4²⁻) depending on the pH of the solution. Under strongly acidic conditions created by sulfuric acid, the dichromate form predominates, while the addition of base shifts the equilibrium toward the chromate form The details matter here..
Applications and Uses in Chemistry
So, the Na2Cr2O7/H2SO4/H2O system has numerous practical applications in both educational and industrial chemistry settings.
Organic Oxidation Reactions
This reagent combination is primarily used for oxidizing organic compounds. The reaction proceeds through the formation of a chromate ester intermediate, which then undergoes elimination to yield the oxidized product. It can convert primary alcohols to carboxylic acids and secondary alcohols to ketones. Take this: ethanol can be oxidized to acetic acid using this reagent system.
Jones Oxidation
One of the most well-known applications is the Jones oxidation, which uses chromic acid generated from sodium dichromate and sulfuric acid to oxidize alcohols. This method is particularly valuable because it selectively oxidizes primary alcohols to carboxylic acids without over-oxidizing most functional groups The details matter here..
Analytical Chemistry
In analytical chemistry, this reagent system is used in the determination of chemical oxygen demand (COD) in water samples. The strong oxidizing power of chromic acid allows it to oxidize nearly all organic compounds, making it useful for assessing water quality and pollution levels That's the part that actually makes a difference..
Surface Cleaning and Preparation
The acidic chromic acid solution is also used for cleaning and passivating glassware in laboratory settings. It effectively removes organic residues and leaves surfaces clean and ready for use in sensitive experiments.
Safety Considerations and Hazards
Working with the Na2Cr2O7/H2SO4/H2O system requires careful attention to safety due to the hazardous nature of the components involved The details matter here..
Chromium(VI) compounds are known carcinogens and pose significant health risks with prolonged exposure. Sodium dichromate and chromic acid are classified as potentially carcinogenic to humans, and inhalation, skin contact, or ingestion should be strictly avoided. Proper personal protective equipment, including gloves, goggles, and lab coats, must be worn at all times when handling these chemicals.
Sulfuric acid is highly corrosive and can cause severe burns upon contact with skin or eyes. It also reacts violently with water, so proper dilution procedures must be followed when preparing solutions Most people skip this — try not to. Less friction, more output..
Proper disposal of chromium-containing solutions is essential due to environmental concerns. Hexavalent chromium is highly toxic to aquatic life and must be treated before being released into the environment. Many laboratories use specialized waste containers for chromium-containing solutions, which are then processed by licensed waste management companies.
The Chemistry Behind the Oxidizing Power
The oxidizing ability of chromic acid stems from the chromium atom in its +6 oxidation state. Chromium(VI) is a powerful electron acceptor, which makes it excellent at removing electrons from organic substrates during oxidation reactions Surprisingly effective..
The mechanism typically involves the formation of a chromate ester between the alcohol and chromic acid. That's why this ester then undergoes a concerted or stepwise elimination process that transfers hydrogen atoms to the chromium center while forming the carbonyl group of the oxidation product. The chromium is simultaneously reduced from Cr(VI) to Cr(III), which is evident in the color change of the solution from orange to green as the reaction progresses Turns out it matters..
This color change serves as a useful visual indicator that oxidation has occurred. The orange color of dichromate ions gives way to the green color of chromium(III) ions, providing a simple way to monitor reaction progress Still holds up..
Frequently Asked Questions
Can I substitute sodium dichromate with potassium dichromate?
Yes, potassium dichromate (K2Cr2O7) can be used similarly to sodium dichromate. The chemistry is essentially the same, with potassium replacing sodium as the counterion. Even so, solubility characteristics may differ slightly between the two salts It's one of those things that adds up..
What is the difference between PCC and this reagent?
Pyridinium chlorochromate (PCC) is a milder version of chromium-based oxidation. While Na2Cr2O7/H2SO4 produces chromic acid in situ, PCC is a pre-formed complex that offers more selective oxidation and is often preferred for sensitive molecules.
Why does the solution change color during oxidation?
The color change from orange to green occurs because chromium(VI) is reduced to chromium(III) during the oxidation process. The orange color comes from dichromate ions, while the green color indicates the presence of chromium(III) ions And it works..
Is this reaction reversible?
The oxidation of organic compounds using chromic acid is generally irreversible, as chromium(III) is the stable reduced form. Still, the equilibrium between chromate and dichromate species in solution is reversible and depends on pH.
Conclusion
The combination of sodium dichromate, sulfuric acid, and water creates a powerful chemical tool that generates chromic acid—a versatile oxidizing agent fundamental to organic synthesis and analytical chemistry. This reagent system enables the oxidation of alcohols to carbonyl compounds, plays a critical role in water quality testing, and serves numerous industrial applications Simple as that..
Understanding what Na2Cr2O7 + H2SO4 + H2O does reveals the elegant chemistry behind chromium(VI)-based oxidations. The transformation from dichromate ions to chromic acid, and ultimately to chromium(III), drives the oxidation of organic substrates while providing visual feedback through characteristic color changes.
People argue about this. Here's where I land on it.
While this reagent system offers tremendous utility in the laboratory, it demands respect for its hazardous nature. Proper safety protocols, including appropriate protective equipment and responsible waste disposal, are essential when working with these powerful chemicals. The combination of powerful oxidizing capability with significant health risks makes this reagent one that should be handled only by those trained in laboratory safety procedures.