The Name Of Fe2o3 Is ....
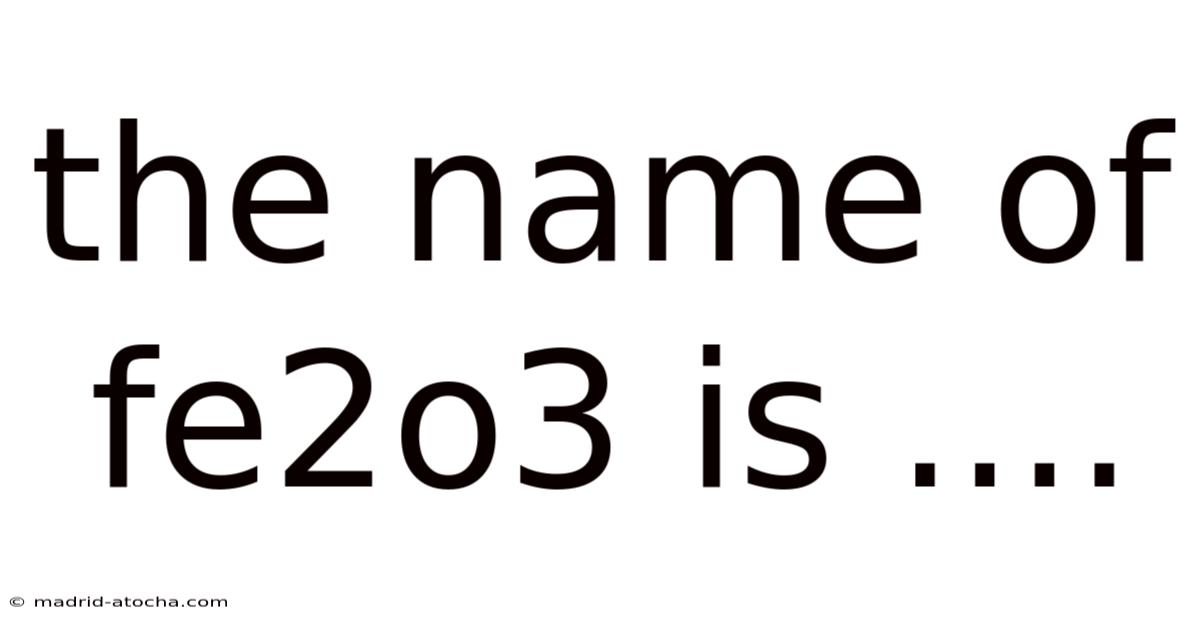
Iron(III) oxide, commonly known as Fe2O3, is an inorganic compound that plays a significant role in various scientific, industrial, and everyday applications. This compound is one of the most abundant iron oxides found in nature and is often recognized for its reddish-brown color. Understanding its properties, structure, and uses is essential for students, researchers, and professionals in chemistry and related fields.
Introduction to Iron(III) Oxide
Iron(III) oxide is the chemical compound with the formula Fe2O3. The name "Iron(III)" refers to the oxidation state of iron in this compound, which is +3. This distinguishes it from other iron oxides such as FeO (iron(II) oxide) and Fe3O4 (magnetite). The Roman numeral III indicates that each iron atom in Fe2O3 has lost three electrons, forming a stable ionic bond with oxygen atoms.
The compound is also known by several other names, including hematite (in its natural mineral form), rust (when referring to the product of iron corrosion), and colcothar (in certain industrial contexts). Its reddish-brown appearance makes it easily recognizable and widely used as a pigment in paints, coatings, and cosmetics.
Chemical Structure and Properties
Fe2O3 has a crystal structure that belongs to the corundum type, where iron atoms are octahedrally coordinated by oxygen atoms. This structure contributes to its stability and various physical properties. The compound is insoluble in water but can dissolve in acids, making it useful in metallurgical processes.
Some key properties of iron(III) oxide include:
- Molar mass: 159.69 g/mol
- Density: 5.24 g/cm³
- Melting point: 1538°C
- Magnetic properties: Weakly ferromagnetic or antiferromagnetic, depending on the crystalline form
These properties make Fe2O3 valuable in both natural and synthetic applications, from being an ore for iron extraction to serving as a catalyst in chemical reactions.
Natural Occurrence and Formation
In nature, Fe2O3 is commonly found as the mineral hematite. Hematite is one of the primary ores of iron and is mined extensively for steel production. The formation of iron(III) oxide can occur through various geological processes, including the oxidation of iron-bearing minerals and the precipitation from iron-rich waters.
In everyday life, iron(III) oxide is often encountered as rust. Rust forms when iron or steel is exposed to oxygen and moisture over time, leading to the oxidation of iron and the formation of Fe2O3 along with other iron oxides. This process is a classic example of corrosion, which has significant implications for engineering and infrastructure maintenance.
Industrial and Practical Applications
Iron(III) oxide is utilized in a wide range of industries due to its unique properties. Some of its major applications include:
- Pigments and dyes: Its red color makes it a popular pigment in paints, coatings, and ceramics.
- Metallurgy: Used as a raw material for iron extraction and steel production.
- Catalysts: Acts as a catalyst in various chemical reactions, including the production of hydrogen and ammonia.
- Magnetic materials: Employed in the manufacture of magnetic recording media and electronic devices.
- Polishing compounds: Used in the form of "jeweler's rouge" for polishing metals and lenses.
These applications highlight the versatility of Fe2O3 and its importance in modern technology and manufacturing.
Scientific and Educational Importance
Understanding iron(III) oxide is crucial for students and professionals in chemistry, materials science, and engineering. The compound serves as an excellent example for studying oxidation states, ionic bonding, and crystal structures. Additionally, its role in corrosion provides practical insights into material durability and protection methods.
In educational settings, Fe2O3 is often used in laboratory experiments to demonstrate chemical reactions, such as the reduction of iron oxides to metallic iron using carbon monoxide or hydrogen. These experiments help students grasp fundamental concepts in chemistry and metallurgy.
Conclusion
Iron(III) oxide, or Fe2O3, is a compound with a rich scientific background and diverse practical applications. Its chemical properties, natural occurrence, and industrial uses make it a subject of ongoing research and importance. Whether encountered as hematite in nature, rust on everyday objects, or a key component in advanced technologies, Fe2O3 continues to play a vital role in both natural and human-made systems. Understanding its characteristics and applications provides valuable insights into the world of chemistry and materials science.
Building on its role incatalysis, Fe₂O₃ also serves as a versatile support material for a host of heterogeneous catalysts. Its high surface area, thermal stability, and ability to host noble‑metal nanoparticles make it indispensable in processes such as selective oxidation, dehydrogenation, and the water‑gas shift reaction. By tuning the particle size and morphology of the iron oxide lattice, researchers can modulate the electronic interaction between the support and the active sites, thereby enhancing turnover frequencies and selectivity. Moreover, doped variants—where transition‑metal ions such as manganese, cobalt, or nickel are introduced into the crystal lattice—have demonstrated superior performance in oxidative coupling of methane and the selective oxidation of alcohols to aldehydes, underscoring the tunability of iron(III) oxide’s catalytic landscape.
Beyond the laboratory, iron(III) oxide nanoparticles have attracted attention for their biomedical potential. Their biocompatibility, magnetic responsiveness, and capacity to generate heat under alternating magnetic fields have positioned them at the forefront of targeted drug delivery, magnetic resonance imaging contrast agents, and photothermal therapy. Surface functionalization with polyethylene glycol or peptide ligands enables these particles to circulate longer in the bloodstream and accumulate preferentially in tumor tissues, while the inherent redox activity of Fe₂O₃ can be harnessed to catalyze the degradation of harmful peroxides within cells, offering a protective shield against oxidative stress.
Environmental considerations also intertwine with the life cycle of iron(III) oxide. In natural waters, Fe₂O₃ colloids can act as scavengers for heavy metals and organic pollutants, facilitating their removal through flocculation and sedimentation. Conversely, the dissolution of iron oxides under acidic conditions can release Fe³⁺ ions, which may influence aquatic chemistry and bioavailability. Understanding these dual roles is essential for designing remediation strategies that leverage the adsorption capacity of iron oxides without inadvertently mobilizing toxic contaminants.
Analytical techniques have evolved to meet the demand for precise quantification of iron(III) oxide in diverse matrices. X‑ray diffraction coupled with Rietveld refinement remains the gold standard for phase identification, while Mössbauer spectroscopy provides a fingerprint of the iron oxidation state and coordination environment. Recent advances in synchrotron‑based X‑ray absorption near‑edge structure (XANES) and X‑ray photoelectron spectroscopy (XPS) enable in‑situ monitoring of surface redox transformations, a critical capability for catalyst design and corrosion studies. These tools collectively empower scientists to probe the atomic‑scale dynamics that dictate macroscopic properties.
Looking forward, the integration of machine‑learning algorithms with high‑throughput experimentation promises to accelerate the discovery of iron‑oxide‑based materials with tailored functionalities. Predictive models can screen thousands of compositions and nanostructures, identifying candidates that exhibit optimal electronic band gaps, magnetic anisotropy, or catalytic turnover numbers. Such data‑driven approaches not only streamline research but also open pathways to sustainable technologies—ranging from efficient solar‑water splitting to low‑temperature CO₂ reduction—where iron(III) oxide plays a pivotal, yet often understated, role.
In sum, iron(III) oxide transcends its humble appearance as rust or pigment. Its rich chemistry, spanning crystal engineering, catalytic versatility, biomedical innovation, and environmental remediation, illustrates a compound that is both timeless and forward‑looking. By continuing to explore its multifaceted behavior through interdisciplinary lenses, scientists and engineers can unlock new applications that harness the full potential of Fe₂O₃, reinforcing its status as a cornerstone of modern material science.
Latest Posts
Latest Posts
-
Are Implants Quarantined Until Results Of Bi Can Be Read
Mar 20, 2026
-
Select The Statement That Best Describes A Feature Of D Snp
Mar 20, 2026
-
Z Value For 99 Confidence Interval
Mar 20, 2026
-
What Are Appropriate Means For Leaving Evidence Of Presence
Mar 20, 2026
-
Complete The Email With The Correct Words In The Box
Mar 20, 2026