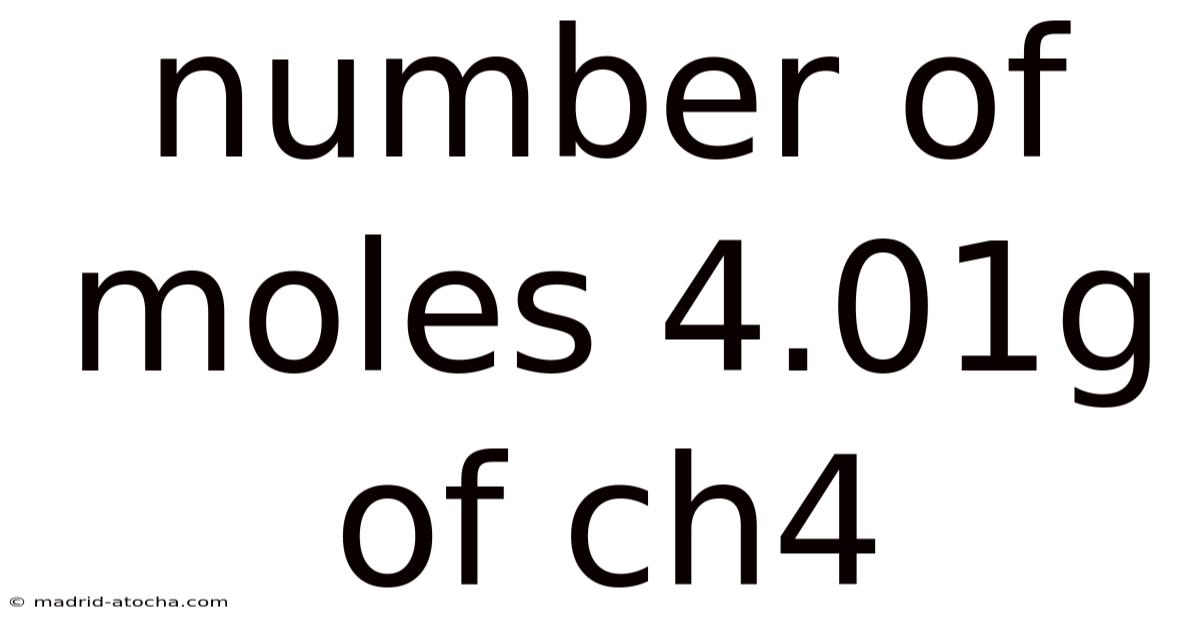To determine the number of moles in 4.And 01 grams of methane (CH₄), we must first understand what a mole represents. A mole is a fundamental unit in chemistry used to count atoms, molecules, or other particles. Plus, just as a dozen means twelve items, a mole represents approximately 6. 022 × 10²³ particles – a number known as Avogadro's constant. This immense quantity allows chemists to work with manageable, measurable amounts of substances while relating macroscopic quantities to microscopic particle counts.
The key to converting between mass (in grams) and moles lies in the molar mass. Now, the molar mass of a substance is the mass of one mole of that substance, expressed in grams per mole (g/mol). It is calculated by summing the atomic masses of all atoms in the chemical formula Worth keeping that in mind..
- Carbon (C) has an atomic mass of approximately 12.01 g/mol.
- Hydrogen (H) has an atomic mass of approximately 1.008 g/mol.
- CH₄ contains 1 carbon atom and 4 hydrogen atoms.
So, the molar mass of CH₄ is: 12.On the flip side, 008 g/mol (H) = 12. 01 g/mol + 4.Also, 032 g/mol = 16. 01 g/mol (C) + 4 × 1.042 g/mol (rounded to three decimal places for precision).
Now, to find the number of moles (n) in a given mass (m), we use the formula: n = m / Molar Mass
Plugging in the values:
- m = 4.01 g
- Molar Mass = 16.042 g/mol
n = 4.01 g / 16.042 g/mol ≈ 0.2499 moles
Which means, 4.Plus, 01 grams of methane (CH₄) contains approximately 0. 25 moles of CH₄ molecules. This calculation is crucial because it allows chemists to translate the mass of a sample into the number of particles it contains, enabling precise stoichiometric calculations in chemical reactions, determining concentrations, and understanding reaction yields And it works..
The mole concept bridges the gap between the tangible world of grams and the abstract world of atoms and molecules. It provides a standardized way to quantify matter at the particle level. Here's a good example: one mole of any substance contains the same number of particles (Avogadro's number), regardless of its chemical identity. This universality makes the mole indispensable for comparing different substances and predicting how they will interact. Without the mole, describing chemical processes quantitatively would be incredibly cumbersome and impractical.
Understanding how to calculate moles from mass is a foundational skill in chemistry. Plus, it underpins virtually every quantitative aspect of the subject, from balancing equations to determining empirical and molecular formulas. Mastering this calculation empowers students and professionals to move smoothly between the laboratory bench and the theoretical framework of chemical principles Not complicated — just consistent..
FAQ
-
Why can't I just use the mass directly without converting to moles?
- Mass alone doesn't tell you how many particles are present. Different substances have different masses per particle (molar masses). Converting mass to moles standardizes the quantity, allowing you to compare different substances or predict reaction amounts based on particle counts.
-
Is the molar mass of CH₄ exactly 16.04 g/mol?
- The atomic masses used (12.01 for C, 1.008 for H) are averages based on the natural abundance of isotopes. While 16.04 g/mol is the standard rounded value, the precise calculation (12.01 + 4*1.008 = 16.042 g/mol) is often used. The difference is negligible for most calculations.
-
How many atoms are in 0.25 moles of CH₄?
- Number of molecules = moles × Avogadro's number = 0.25 × 6.022 × 10²³ = 1.5055 × 10²³ molecules.
- Each CH₄ molecule contains 5 atoms (1 C + 4 H). So, total atoms = 1.5055 × 10²³ molecules × 5 atoms/molecule = 7.5275 × 10²³ atoms.
-
Why is the mole concept important?
- It provides a practical way to count particles using measurable mass. It allows chemists to write balanced chemical equations, calculate reactant requirements, predict product yields, and determine concentrations (molarity) – all essential for research, industry, and medicine.
-
Can I use this method for any compound?
- Absolutely. The method (moles = mass / molar mass) is universal. You simply need the correct molar mass for the specific compound, calculated from its chemical formula.
Conclusion
Calculating the number of moles in a given mass of substance is a fundamental and indispensable skill in chemistry. For 4.Plus, 01 grams of methane (CH₄), with a molar mass of approximately 16. 042 grams per mole, the calculation yields 0.2499 moles, which rounds to 0.25 moles. In practice, this conversion bridges the tangible world of grams with the abstract world of atoms and molecules, enabling precise quantitative analysis and prediction in chemical processes. Mastery of this concept is essential for any student or professional navigating the quantitative landscape of chemistry, providing the foundation for understanding reaction stoichiometry, solution concentrations, and material properties. The mole, though seemingly abstract, is a powerful tool that makes the invisible world of particles measurable and predictable Simple, but easy to overlook. Took long enough..
serves as the critical link connecting the laboratory bench and the theoretical framework of chemical principles. By translating measurable mass into proportional particle counts, chemists can confidently scale reactions from microscopic test tubes to industrial reactors. This proportional reasoning extends far beyond simple unit conversions; it underpins limiting reactant analysis, percent yield calculations, and gas law applications, where macroscopic properties like pressure and volume are directly tied to molar quantities Most people skip this — try not to..
When applying these calculations in practice, attention to detail ensures accuracy. Always retain extra significant figures through intermediate steps to prevent compounding rounding errors, and verify that your chemical formula accurately reflects the compound’s true composition—particularly when working with hydrates, isotopically labeled compounds, or complex coordination species. What's more, maintain consistency in your reference data, as slight variations in standard atomic weights across different periodic tables can affect high-precision analytical work. Documenting your molar mass sources and calculation steps not only improves reproducibility but also strengthens overall experimental integrity.
Conclusion
Mastering the mass-to-mole conversion transforms abstract chemical formulas into actionable, measurable reality. It stands as the cornerstone of quantitative chemistry, enabling precise dosage formulation in pharmaceuticals, accurate pollutant tracking in environmental science, and efficient process scaling in industrial manufacturing. Beyond the arithmetic, this skill cultivates a proportional mindset essential for interpreting reaction dynamics, predicting material behavior, and troubleshooting experimental discrepancies. With this foundational tool securely in place, the quantitative challenges of chemistry cease to be obstacles and instead become reliable, predictable pathways to scientific discovery and real-world innovation.
Beyond manual documentation and static calculations, contemporary chemical practice increasingly embeds molar relationships into automated workflows and digital modeling environments. Laboratory information management systems, real-time process analytics, and computational chemistry platforms routinely translate mass inputs into molar equivalents, streamlining everything from high-throughput compound screening to continuous-flow manufacturing. Yet, reliance on algorithmic precision does not diminish the necessity of conceptual fluency. Software can execute the arithmetic, but only a trained practitioner can contextualize statistical outliers, recognize when a deviation points to hidden impurities or competing reaction pathways, and recalibrate experimental parameters accordingly. This synergy between computational efficiency and foundational understanding ensures that quantitative rigor remains intact even as laboratory technologies rapidly evolve.
The pedagogical approach to teaching these relationships has similarly matured, shifting away from rote memorization toward dynamic, visualization-driven problem solving. In real terms, interactive molecular simulations, virtual reaction modules, and data-rich case studies allow learners to observe directly how particle-level proportions manifest in macroscopic outcomes. This immersive training builds the intuitive grasp necessary for tackling next-generation challenges, such as designing atom-economical syntheses, engineering catalytic cycles with minimal waste, or calibrating sensors for trace environmental analytes. In each scenario, the ability to move fluidly between measurable quantities and molecular counts dictates both the feasibility and the long-term sustainability of the work But it adds up..
Conclusion
At its core, the mass-to-mole conversion is the operational heartbeat of quantitative chemistry, transforming static formulas into dynamic, experimentally verifiable frameworks. As the discipline advances toward increasingly complex, data-intensive, and sustainability-driven paradigms, this foundational competency will only grow in relevance. So it empowers scientists to handle the full spectrum of chemical inquiry—from synthesizing novel materials with exacting precision to monitoring ecological systems with analytical confidence. By internalizing the relationship between tangible mass and molecular quantity, chemists equip themselves not merely to calculate, but to comprehend, innovate, and responsibly shape the material world.