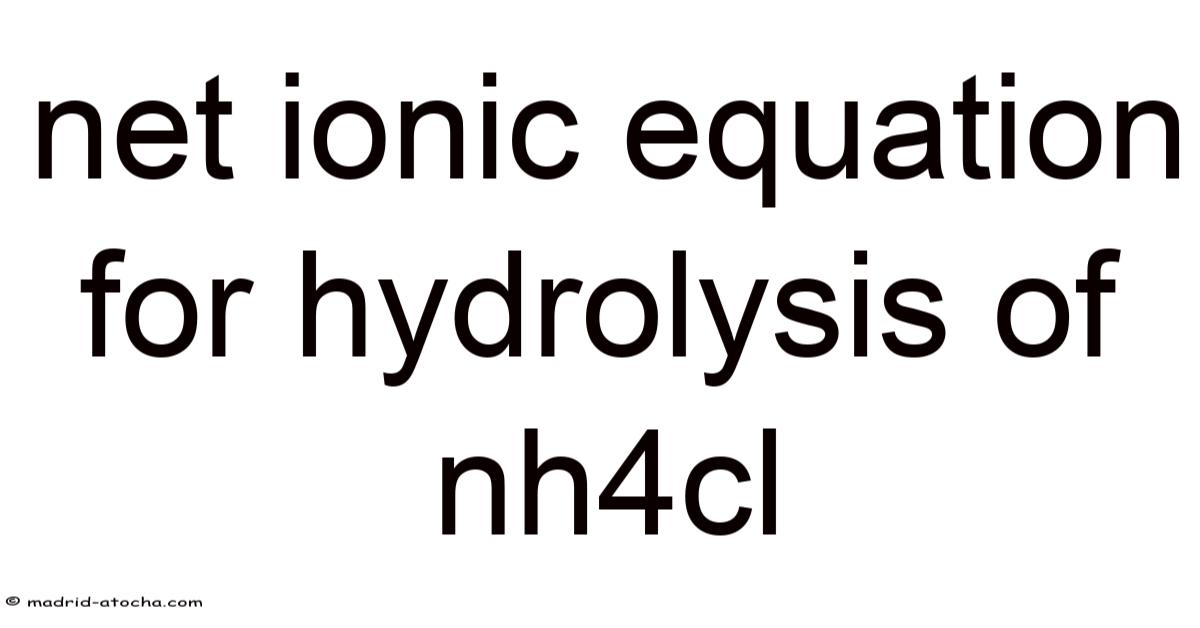Introduction
The net ionic equation for hydrolysis of NH4Cl is a fundamental concept in acid‑base chemistry that explains how an apparently neutral salt can generate an acidic solution. When ammonium chloride (NH₄Cl) dissolves in water, the cation NH₄⁺ reacts with water to produce hydronium ions (H₃O⁺) while the anion Cl⁻ remains unchanged. Understanding this net ionic equation helps students predict pH, interpret titration curves, and apply the principles of equilibrium to real‑world problems. This article breaks down the process step by step, provides the balanced net ionic equation, and answers common questions to reinforce learning.
Steps to Derive the Net Ionic Equation
-
Write the complete ionic equation
- Dissolve NH₄Cl:
[ \text{NH}_4\text{Cl (s)} \rightarrow \text{NH}_4^+ \text{ (aq)} + \text{Cl}^- \text{ (aq)} ] - Water does not appear as a reactant in the complete ionic form because it is the solvent.
- Dissolve NH₄Cl:
-
Identify the species that actually participate in the reaction
- NH₄⁺ reacts with water (H₂O) to form NH₃ and H₃O⁺.
- Cl⁻ is a spectator ion (it does not change chemically) and therefore does not appear in the net ionic equation.
-
Write the chemical reaction of NH₄⁺ with water
[ \text{NH}_4^+ + \text{H}_2\text{O} \rightleftharpoons \text{NH}_3 + \text{H}_3\text{O}^+ ] -
Combine with the spectator ion (optional for full molecular equation)
- Add Cl⁻ back to both sides to maintain mass balance:
[ \text{NH}_4^+ + \text{Cl}^- + \text{H}_2\text{O} \rightleftharpoons \text{NH}_3 + \text{H}_3\text{O}^+ + \text{Cl}^- ]
- Add Cl⁻ back to both sides to maintain mass balance:
-
Cancel spectator ions (Cl⁻) to obtain the net ionic equation:
[ \boxed{\text{NH}_4^+ + \text{H}_2\text{O} \rightleftharpoons \text{NH}_3 + \text{H}_3\text{O}^+} ]
This concise representation captures the essential chemistry of the hydrolysis process It's one of those things that adds up..
Scientific Explanation
What Is Hydrolysis?
Hydrolysis is the reaction of an ion (or molecule) with water that results in the formation of a new species and the release or consumption of a proton (H⁺). In the case of NH₄⁺, the cation acts as a weak acid because it can donate a proton to water, generating hydronium ions and leaving behind a weak base (NH₃).
Equilibrium Constant (Kₐ)
The acid‑dissociation constant for NH₄⁺ is derived from the ion‑product of water (K_w) and the base‑dissociation constant of ammonia (K_b):
[ K_a = \frac{K_w}{K_b} ]
At 25 °C, (K_w = 1.Here's the thing — 0 \times 10^{-14}) and (K_b \approx 1. Even so, 6 \times 10^{-10}). 8 \times 10^{-5}), giving (K_a \approx 5.A small Kₐ indicates that the reaction proceeds only partially, which explains why a solution of NH₄Cl is slightly acidic rather than strongly acidic Less friction, more output..
Role of Water
Water acts both as a solvent and as a reactant. In the net ionic equation, water is the source of the hydroxide portion that accepts the proton from NH₄⁺, forming H₃O⁺. This is why the reaction is written with H₂O on the reactant side Simple, but easy to overlook..
pH Implications
Because the net ionic equation produces H₃O⁺, the concentration of hydronium ions increases, lowering the pH. For a 0.10 M NH₄Cl solution, the calculated pH is typically around 5.1, confirming the acidic nature of the hydrolysis That alone is useful..
Frequently Asked Questions (FAQ)
1. Why is Cl⁻ considered a spectator ion?
Cl⁻ does not undergo any chemical change during the hydrolysis of NH₄Cl; it simply remains dissolved as an anion. Spectator ions appear on both sides of the complete ionic equation and cancel out when the net ionic equation is written Nothing fancy..
2. Can the net ionic equation be written without water?
No. Water is essential as the medium that accepts the proton. Omitting H₂O would incorrectly suggest that NH₄⁺ dissociates spontaneously into NH₃ and H⁺, which is not chemically accurate.
3. How does temperature affect the hydrolysis of NH₄Cl?
Increasing temperature generally increases the value of K_w and K_b, which in turn influences Kₐ. The net ionic equation remains the same, but the equilibrium position may shift, leading to a slightly different pH.
4. Is the net ionic equation applicable to all ammonium salts?
Yes, for any ammonium salt (e.g., NH₄NO₃, NH₄SO₄), the cation NH₄⁺ will undergo the same hydrolysis reaction, while the accompanying anion determines the additional ionic strength but does not participate in the net ionic equation Most people skip this — try not to..
5. What would happen if we added a strong base to the solution?
Adding a strong base (OH⁻) would consume H₃O⁺, shifting the equilibrium toward the left (Le Chatelier’s principle). This would reduce the acidity and drive the reaction to produce more NH₃, eventually neutralizing the solution.
Conclusion
The net ionic equation for hydrolysis of NH4Cl — (\text{NH}_4^+ + \text{H}_2\text{O} \rightleftharpoons \text{NH}_3 + \text{H}_3\text{O}^+) — succinctly captures the essential chemistry that makes an apparently neutral salt produce an acidic solution. By recognizing NH₄⁺ as a weak acid and Cl⁻ as a spectator ion, students can easily derive the equation, understand the underlying equilibrium, and predict the resulting pH. Mastery of this concept not only supports academic success in chemistry courses but also provides a foundation for interpreting real‑world phenomena such as soil acidity, ocean buffering, and industrial chemical processes.
The hydrolysis of NH₄Cl illustrates a fundamental principle in chemistry: the behavior of ions in solution can dramatically alter the properties of compounds. By focusing on the net ionic equation, we simplify the complexity of the reaction and highlight the key players — NH₄⁺ and H₂O — while acknowledging the role of spectator ions like Cl⁻. This approach not only aids in understanding the specific case of NH₄Cl but also provides a framework for analyzing similar reactions involving other ammonium salts Which is the point..
Adding to this, the pH implications of this reaction underscore the importance of acid-base equilibria in determining the acidity or basicity of solutions. Day to day, the calculated pH for a 0. Which means 10 M NH₄Cl solution, around 5. Practically speaking, 1, is a direct consequence of the increased concentration of H₃O⁺ ions resulting from the hydrolysis of NH₄⁺. This practical example reinforces the theoretical concepts of acid strength, equilibrium constants, and the impact of ion concentration on pH.
The FAQs provided offer a full breakdown to common questions and misconceptions surrounding the hydrolysis of NH₄Cl, making the topic more accessible to students and learners. By addressing these questions, we not only clarify the chemistry but also underline the broader principles of chemical equilibrium, the role of spectator ions, and the influence of environmental factors such as temperature on chemical reactions.
All in all, the hydrolysis of NH₄Cl serves as an excellent case study for exploring the principles of acid-base chemistry and the behavior of ions in solution. Through a deep dive into the net ionic equation, pH implications, and related FAQs, we gain a comprehensive understanding of this reaction and its broader implications in chemistry. This knowledge is not only academically valuable but also essential for interpreting and predicting chemical behavior in various real-world contexts, from environmental science to industrial applications. Mastery of these concepts empowers students to tackle more complex chemical systems with confidence and insight.