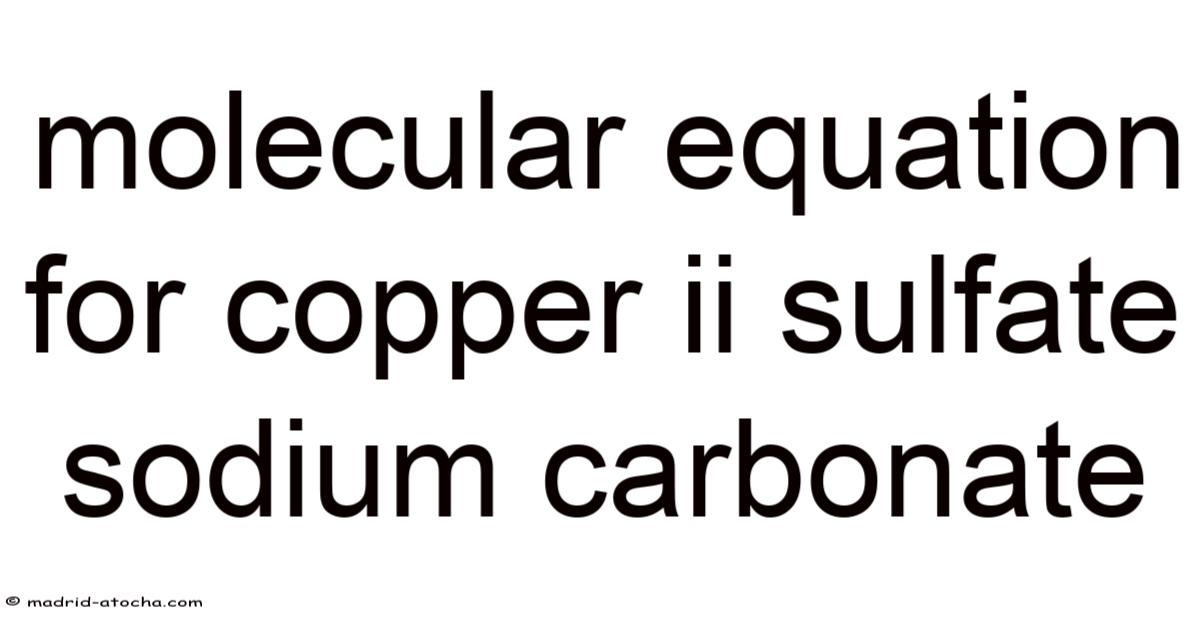Introduction
Copper II sulfate reacts with sodium carbonate to produce a characteristic blue precipitate of copper carbonate while releasing sodium sulfate into the solution, illustrating a classic double displacement (metathesis) reaction in aqueous chemistry. This transformation is frequently demonstrated in school laboratories because the visual change—formation of a vivid blue solid—provides an immediate, observable confirmation of the reaction’s occurrence. Understanding the molecular equation, the ionic equation, and the net ionic equation for this process deepens comprehension of how ions exchange partners, how solubility rules dictate product formation, and how spectator ions can be eliminated to reveal the essential chemical change Easy to understand, harder to ignore..
Balanced Molecular Equation
The complete balanced molecular equation for the reaction between copper II sulfate and sodium carbonate is:
CuSO₄ (aq) + Na₂CO₃ (aq) → CuCO₃ (s) + Na₂SO₄ (aq)
Key points:
- CuSO₄ is copper II sulfate, a soluble ionic compound.
- Na₂CO₃ is sodium carbonate, also soluble.
- CuCO₃ precipitates because copper carbonate is poorly soluble in water.
- Na₂SO₄ remains dissolved as sodium sulfate, a highly soluble salt.
Balancing the equation ensures that the number of each type of atom is equal on both sides, satisfying the law of conservation of mass.
Ionic Equation
When the compounds dissolve in water, they dissociate into their constituent ions. Writing the ionic equation reveals the actual species that participate in the reaction:
Cu²⁺ (aq) + SO₄²⁻ (aq) + 2 Na⁺ (aq) + CO₃²⁻ (aq) → CuCO₃ (s) + 2 Na⁺ (aq) + SO₄²⁻ (aq)
In this equation, the sulfate ion (SO₄²⁻) and the sodium ion (Na⁺) appear unchanged on both sides, indicating that they are spectator ions—they do not undergo any chemical change.
Net Ionic Equation
By canceling the spectator ions from the ionic equation, we obtain the net ionic equation, which captures the essence of the chemical transformation:
Cu²⁺ (aq) + CO₃²⁻ (aq) → CuCO₃ (s)
This simplified representation shows that the copper(II) cation directly combines with carbonate anion to form solid copper carbonate.
Scientific Explanation
Solubility and Precipitation
The driving force behind this reaction is the difference in solubility between the reactants and products. Copper II sulfate and sodium carbonate are both highly soluble in water, meaning their ions are readily dispersed in solution. Copper carbonate, however, has a low solubility product (K_sp), causing it to precipitate out of the solution once the product of the ion concentrations exceeds its K_sp threshold That's the whole idea..
Double Displacement Mechanism
This reaction exemplifies a double displacement mechanism, where the cations and anions swap partners:
- Copper(II) (Cu²⁺) pairs with carbonate (CO₃²⁻) to form copper carbonate.
- Sodium (Na⁺) pairs with sulfate (SO₄²⁻) to form sodium sulfate.
The precipitation of copper carbonate is a visual cue that the reaction has proceeded, while sodium sulfate remains in solution, invisible to the naked eye.
Factors Influencing the Reaction
- Concentration of Reactants – Higher molar concentrations increase the likelihood that the ion product will surpass the solubility limit, leading to faster and more complete precipitation.
- Temperature – Elevated temperatures generally increase the solubility of ionic compounds, which can reduce precipitation efficiency; cooling the solution often enhances the formation of the blue precipitate.
- Presence of Other Ions – Adding competing ions (e.g., excess Na⁺ or SO₄²⁻) can shift the equilibrium, either suppressing or promoting the precipitation of copper carbonate depending on the common‑ion effect.
Safety and Practical Considerations
- Copper II sulfate is an irritant; handle it with gloves and avoid inhalation of dust.
- Sodium carbonate (washing soda) is mildly alkaline and can cause skin irritation; use protective eyewear.
- The reaction generates copper carbonate, which is not highly toxic but should be treated as a chemical
Applications and Observations
Copper(II) carbonate, the striking blue precipitate formed in this reaction, has several practical uses. In modern times, it is employed as a fungicide in agriculture, where its low solubility allows for sustained release of copper ions that inhibit fungal growth. Think about it: historically, it served as a pigment in paints and ceramics, prized for its vivid color. Additionally, copper carbonate acts as a precursor in the synthesis of other copper compounds, such as copper(II) oxide, which is used in catalysis and electronic materials.
Short version: it depends. Long version — keep reading.
In the laboratory, this reaction is a classic demonstration for teaching solubility rules and precipitation concepts. The immediate appearance of the blue solid provides a clear visual indicator of a chemical change, making it an effective educational tool. Students can also explore variables such as reactant concentration and temperature by observing how these factors affect the quantity and texture of the precipitate.
And yeah — that's actually more nuanced than it sounds Not complicated — just consistent..
Broader Chemical Context
This reaction is a specific example of a double displacement that leads to an insoluble product, a common pattern in aqueous chemistry. So it underscores fundamental principles such as the role of solubility products in predicting precipitation and the conservation of mass in ionic equations. On top of that, it illustrates Le Chatelier’s principle: adding excess carbonate ions, for instance, can shift the equilibrium to produce more precipitate, while adding acid dissolves it by reacting with carbonate to form carbon dioxide and water.
Understanding such reactions is crucial in fields like environmental chemistry, where precipitation is used to remove heavy metals from wastewater, and in analytical chemistry, where selective precipitation aids in identifying ions in solution That's the part that actually makes a difference..
Conclusion
The reaction between copper(II) sulfate and sodium carbonate is more than a simple classroom demonstration—it is a window into core chemical principles. By forming copper(II) carbonate precipitate, it visually reinforces concepts of solubility, ionic interactions, and equilibrium. The net ionic equation, Cu²⁺ (aq) + CO₃²⁻ (aq) → CuCO₃ (s), distills the reaction to its essence, highlighting the direct combination of ions to create a new substance Worth knowing..
Beyond theory, this reaction has tangible applications in pigments, agriculture, and industry, demonstrating how basic chemical processes underpin practical technologies. Whether used to educate budding chemists or to produce useful materials, the formation of copper(II) carbonate remains a compelling example of chemistry in action. By studying such reactions, we gain both knowledge and appreciation for the complex dance of ions that shapes the material world.
Extending the Investigation#### 1. Quantitative Aspects and Stoichiometry
When the solutions are mixed in precisely stoichiometric proportions, the theoretical yield of CuCO₃ can be calculated from the limiting reagent. For a 0.020 mol L⁻¹ CuSO₄ solution combined with an equimolar Na₂CO₃ solution, the maximum mass of precipitate expected is
[ m_{\text{CuCO}3}= n{\text{Cu}^{2+}}\times M_{\text{CuCO}_3}=0.020;\text{mol}\times 123.55;\text{g mol}^{-1}=2.47;\text{g} ]
In practice, the isolated solid often weighs slightly less because of entrapped solution and incomplete drying. In real terms, by weighing the dried precipitate before and after washing with a small volume of deionized water, students can perform a gravimetric analysis that reinforces concepts of percent yield and sources of experimental error (e. g., incomplete precipitation, hygroscopic nature of CuCO₃).
2. Kinetic Observations
The precipitation is essentially instantaneous; the blue solid appears within seconds of mixing. On the flip side, subtle kinetic differences become evident when the temperature is varied. Raising the mixture from 20 °C to 40 °C accelerates the formation of nuclei, leading to finer particles that settle more slowly. Conversely, cooling the reaction to 5 °C yields larger, more rapidly sedimenting crystals. Such temperature dependence offers a tangible demonstration of how molecular motion influences nucleation rates.
3. Surface Morphology and Its Consequences
Scanning electron microscopy (SEM) reveals that the precipitate consists of irregular, plate‑like crystals whose surfaces are riddled with nanoscale pores. This morphology increases the effective surface area, which in turn enhances the material’s reactivity in subsequent transformations—such as its conversion to CuO upon heating. The porous texture also explains why the solid adsorbs dyes more efficiently than a dense, crystalline counterpart, a property exploited in pigment formulation Not complicated — just consistent. That alone is useful..
4. Analytical Applications
Because CuCO₃ precipitates selectively in the presence of carbonate under neutral to slightly basic conditions, it serves as a qualitative test for copper(II) ions in complex matrices. In wastewater treatment, a controlled addition of sodium carbonate can co‑precipitate copper along with other heavy metals, simplifying downstream removal steps. On top of that, the stoichiometric relationship between Cu²⁺ and carbonate permits the development of titration protocols where the endpoint is signaled by the sudden appearance of a blue turbidity, offering a low‑cost alternative to instrumental analysis.
5. Environmental and Safety Considerations
While CuCO₃ is only sparingly soluble, it can still contribute to copper accumulation in aquatic ecosystems if released in large quantities. Regulatory frameworks therefore mandate that effluents containing copper be treated to meet permissible concentrations (often <1 mg L⁻¹). Laboratory practitioners are advised to neutralize any residual acidity before disposal and to collect the precipitate for proper disposal according to hazardous waste protocols. The reaction also generates carbon dioxide when acid is added to dissolve the precipitate, a reminder of the interconnectedness of acid‑base and carbonate chemistry.
6. Historical Perspective
The first documented synthesis of copper carbonate dates back to the 18th century, when alchemists sought to produce verdigris—a green pigment derived from the oxidation of copper. Their methods inadvertently produced the same blue‑green solid that modern chemists now isolate through the simple mixing of copper sulfate and sodium carbonate. This historical link underscores how contemporary laboratory techniques echo centuries‑old practices, bridging the gap between artisanal pigment making and today’s precision analytical chemistry.
Final Conclusion
The seemingly elementary reaction between copper(II) sulfate and sodium carbonate unfolds as a rich tapestry of chemical principles, from solubility equilibria and ionic interactions to kinetic nuances and practical applications. Day to day, by transforming dissolved ions into a vivid blue precipitate, the process makes abstract concepts tangible, offering educators a powerful visual tool and researchers a versatile reagent for pigment production, metal recovery, and analytical testing. The net ionic equation, Cu²⁺ (aq) + CO₃²⁻ (aq) → CuCO₃ (s), distills the essence of the transformation, yet the surrounding layers of observation—temperature effects, crystal morphology, quantitative yields, and environmental implications—enrich the narrative far beyond a single line of symbols.
In appreciating this reaction, we gain more than a demonstration of precipitation; we glimpse how fundamental chemistry
7. Analytical Extensions and Modern Variations
7.1 Spectroscopic Confirmation
Although the blue hue of copper(II) carbonate is unmistakable to the naked eye, quantitative confirmation can be achieved with UV‑Vis spectroscopy. A dilute suspension of the precipitate exhibits a broad absorption band centered around 620 nm, corresponding to d‑d transitions in the Cu²⁺ ion. By measuring the absorbance of the supernatant before and after precipitation, one can calculate the percentage of copper removed using Beer‑Lambert law, providing a rapid check on reaction completeness without resorting to gravimetric weighing Took long enough..
7.2 Microscopic Characterization
Scanning electron microscopy (SEM) coupled with energy‑dispersive X‑ray spectroscopy (EDS) reveals the morphology and elemental composition of the precipitate. Typical CuCO₃ crystals appear as irregular, plate‑like aggregates ranging from 2–10 µm in size. EDS spectra confirm the presence of copper, carbon, and oxygen in the expected stoichiometric ratios, while the absence of sodium or sulfate peaks validates the purity of the product after thorough washing.
7.3 Green Chemistry Adaptations
In line with the 12 Principles of Green Chemistry, recent work has explored the use of biogenic carbonate sources (e.g., calcium carbonate derived from shell waste) and copper‑rich industrial effluents as feedstocks. By replacing sodium carbonate with calcium carbonate, the by‑product becomes calcium sulfate, which can be recovered and reused in the cement industry, thereby closing material loops and reducing the overall environmental footprint Not complicated — just consistent..
7.4 Electrochemical Recovery
An emerging technique couples the precipitation step with an electrochemical cell that applies a modest cathodic potential (≈ ‑0.2 V vs. Ag/AgCl) to the suspension. The electric field promotes nucleation on the cathode surface, yielding a denser, adherent CuCO₃ film that can be directly harvested. This method minimizes the need for filtration and reduces waste water generation, offering a scalable route for copper reclamation from mining leachates.
8. Troubleshooting Guide
| Symptom | Likely Cause | Remedy |
|---|---|---|
| No visible precipitate | Insufficient carbonate concentration or pH < 5 | Add a calculated excess of Na₂CO₃ and verify pH; gently warm the mixture to 30‑35 °C to increase carbonate ion availability. |
| Precipitate dissolves on standing | Formation of basic copper carbonate (malachite) due to excess hydroxide | Adjust pH to 6–7 using dilute HCl; avoid adding strong bases before precipitation. |
| Chalky, white precipitate | Presence of calcium or magnesium impurities forming carbonate salts | Use high‑purity reagents; pre‑filter solutions through ion‑exchange resins to remove divalent cations. |
| Fine, colloidal suspension that does not settle | Rapid addition of reagents causing nucleation of nano‑particles | Add reagents slowly with vigorous stirring; employ a flocculant (e.g.In practice, , polyacrylamide) to aggregate particles. |
| Persistent blue tint in filtrate | Incomplete precipitation due to low ionic strength | Increase overall ionic strength by adding a neutral salt (e.g., NaCl) to promote salting‑out effect. |
9. Pedagogical Integration
Educators can embed this reaction into a multiphase laboratory module:
- Pre‑lab discussion – Review solubility product constants (Ksp) and Le Chatelier’s principle.
- Hands‑on experiment – Students prepare a series of mixtures varying Na₂CO₃ concentration, record precipitate mass, and plot % copper removal versus carbonate excess.
- Data analysis – Fit the experimental data to a Langmuir‑type adsorption isotherm to illustrate surface‑controlled precipitation.
- Reflection – Connect findings to real‑world scenarios such as wastewater treatment, pigment manufacturing, and historical art conservation.
This scaffolded approach reinforces quantitative reasoning while maintaining the visual appeal that makes the copper carbonate precipitation a perennial favorite in introductory chemistry labs Worth keeping that in mind..
Concluding Remarks
The precipitation of copper(II) carbonate from copper(II) sulfate and sodium carbonate is far more than a textbook example of an insoluble salt forming. It serves as a nexus where thermodynamics, kinetics, analytical techniques, environmental stewardship, and historical context converge. The reaction’s simplicity enables rapid visual confirmation, yet its depth offers ample avenues for advanced investigation—spectroscopic validation, microscopic morphology studies, green‑chemistry process redesign, and electrochemical integration No workaround needed..
By mastering this system, students and practitioners alike gain a concrete illustration of how manipulating ion concentrations and pH can drive selective separation, a principle that underlies countless industrial and environmental processes. On top of that, the awareness of safety and disposal considerations ensures that the laboratory experience remains responsible and sustainable.
In essence, the blue precipitate that settles at the bottom of the beaker is a small but vivid reminder: even the most straightforward chemical transformation can open a window onto a broad landscape of scientific inquiry and practical application.