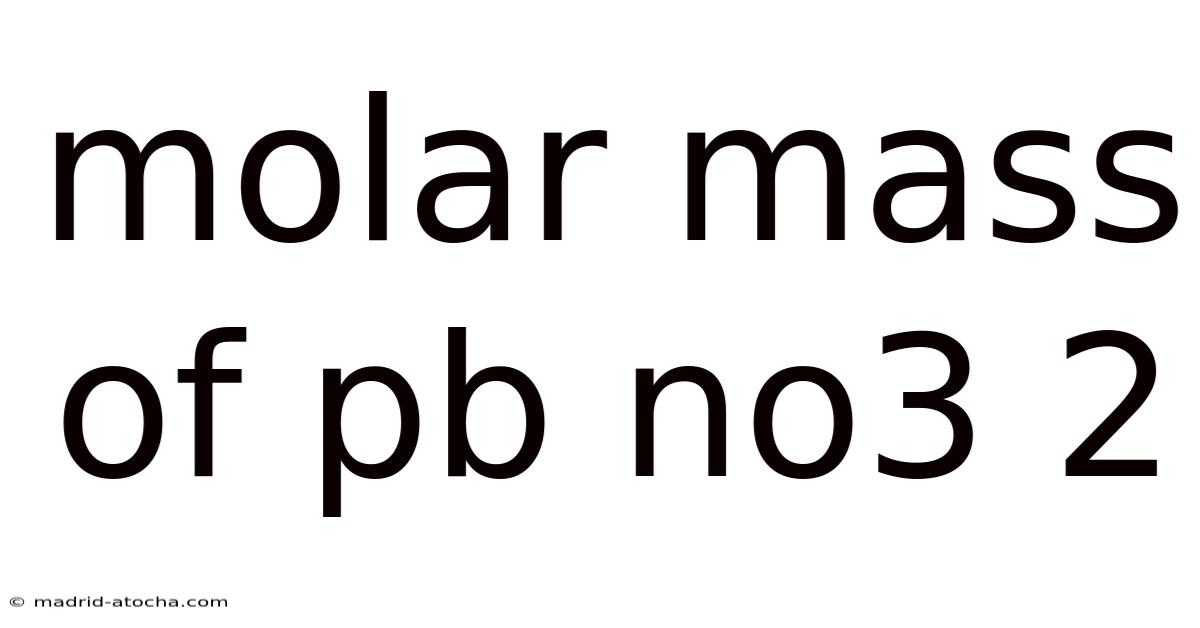Understanding the Molar Mass of Pb(NO3)2: Lead(II) Nitrate
The molar mass of Pb(NO3)2, also known as lead(II) nitrate, is a fundamental concept in chemistry that helps scientists and students understand the quantitative relationships in chemical reactions. This compound, consisting of lead cations and nitrate anions, has significant applications in various fields, from laboratory experiments to industrial processes. Calculating the molar mass of Pb(NO3)2 requires understanding the atomic composition of the compound and applying the periodic table values accurately. In this full breakdown, we'll explore how to determine the molar mass of Pb(NO3)2, its significance in chemistry, and practical applications that demonstrate its importance.
What is Pb(NO3)2?
Lead(II) nitrate, with the chemical formula Pb(NO3)2, is an inorganic compound composed of one lead atom (Pb) and two nitrate groups (NO3). The Roman numeral II in the name indicates that lead in this compound has an oxidation state of +2. Which means the nitrate group consists of one nitrogen atom and three oxygen atoms. This white crystalline solid is highly soluble in water and has been historically used in various applications, including matches and fireworks, though its use has declined due to lead's toxicity Easy to understand, harder to ignore..
This is the bit that actually matters in practice.
The molecular structure of Pb(NO3)2 features a central lead ion surrounded by two nitrate ions, with each nitrate ion having a trigonal planar geometry. The compound's physical properties, such as solubility and reactivity, are directly related to its molar mass, which determines how much of the substance can be dissolved in a given volume or how it will behave in chemical reactions.
Step-by-Step Calculation of Molar Mass
To calculate the molar mass of Pb(NO3)2, we need to follow a systematic approach using the periodic table:
-
Identify the elements present: The compound contains lead (Pb), nitrogen (N), and oxygen (O).
-
Determine the quantity of each atom:
- Lead (Pb): 1 atom
- Nitrogen (N): 2 atoms (since there are two NO3 groups)
- Oxygen (O): 6 atoms (2 nitrate groups × 3 oxygen atoms each)
-
Find the atomic masses (from the periodic table):
- Lead (Pb): 207.2 g/mol
- Nitrogen (N): 14.01 g/mol
- Oxygen (O): 16.00 g/mol
-
Calculate the total mass for each element:
- Lead: 1 × 207.2 = 207.2 g/mol
- Nitrogen: 2 × 14.01 = 28.02 g/mol
- Oxygen: 6 × 16.00 = 96.00 g/mol
-
Add the masses together:
- Total molar mass = 207.2 + 28.02 + 96.00 = 331.22 g/mol
Because of this, the molar mass of Pb(NO3)2 is 331.And 22 g/mol. Plus, this value represents the mass of one mole of lead(II) nitrate, which contains Avogadro's number (6. 022 × 10^23) of Pb(NO3)2 formula units Simple as that..
Scientific Explanation of Molar Mass
Molar mass is defined as the mass of one mole of a substance, expressed in grams per mole (g/mol). Consider this: it is essentially the weight of all the atoms in a molecule or formula unit. The concept of molar mass bridges the gap between the microscopic world of atoms and molecules and the macroscopic world that we can measure in the laboratory Worth keeping that in mind..
For Pb(NO3)2, understanding its molar mass requires recognizing that it's an ionic compound formed between Pb²⁺ ions and NO₃⁻ ions. The crystal structure of solid Pb(NO3)2 consists of these ions arranged in a lattice, but when we calculate molar mass, we consider the formula unit as the smallest repeating unit of the compound Worth knowing..
The molar mass calculation is based on the atomic masses of the constituent elements, which are weighted averages of the masses of all naturally occurring isotopes of each element. Here's the thing — for example, lead has several isotopes, with Pb-207 being the most abundant, which contributes to its atomic mass of 207. 2 g/mol That's the part that actually makes a difference. Nothing fancy..
Applications and Significance
The molar mass of Pb(NO3)2 has numerous practical applications in chemistry and related fields:
-
Stoichiometric calculations: In chemical reactions involving Pb(NO3)2, the molar mass allows chemists to calculate the exact amounts of reactants needed and products formed. Here's one way to look at it: in the reaction between Pb(NO3)2 and KI to form PbI2 precipitate, knowing the molar mass enables precise measurements.
-
Solution preparation: When preparing solutions of Pb(NO3)2, chemists use the molar mass to determine how much solid to dissolve to achieve a desired molarity. A 1 M solution of Pb(NO3)2 would contain 331.22 grams of the compound per liter of solution.
-
Analytical chemistry: In gravimetric analysis, the molar mass is essential for calculating the amount of a substance based on the mass of a precipitate. To give you an idea, if PbI2 is precipitated from a solution containing Pb(NO3)2, the molar mass of both compounds allows quantification of the original lead content.
-
Industrial processes: In manufacturing, understanding the molar mass helps in scaling up reactions from laboratory to industrial production, ensuring the correct proportions of materials are used And it works..
-
Environmental monitoring: Due to lead's toxicity, environmental agencies monitor lead levels in water and soil. The molar mass of compounds like Pb(NO3)2 is used to convert between different units of concentration when assessing contamination.
Common Mistakes to Avoid
When calculating the molar mass of Pb(NO3)2, several common errors can occur:
-
Incorrect atom counting: Forgetting that there are two nitrate groups (NO3) and thus two nitrogen atoms and six oxygen atoms is a frequent mistake. Always use parentheses to identify groups that repeat.
-
Using outdated atomic masses: Atomic masses on the periodic table can be updated as measurement techniques improve. Always use the most current values available Still holds up..
-
Ignoring significant figures: The final answer should reflect the precision of the atomic masses used. For Pb(NO3)2, the molar mass is typically reported to two decimal places (331.22 g/mol) Most people skip this — try not to..
-
Confusing molecular mass with molar mass: Molecular mass (or molecular weight) is the mass of a single molecule expressed in atomic mass units (amu), while molar mass is the mass of one mole of molecules expressed in g/mol.
-
Misinterpreting chemical formulas: Ensure you correctly interpret the formula Pb(NO3)2, which indicates two nitrate groups, not PbN6O6, which would represent a different compound Nothing fancy..
Frequently Asked Questions
Q: Why is the molar mass of Pb(NO3)2 important in chemistry? A: The molar mass is crucial for stoichiometric calculations, solution preparation, and analytical procedures. It allows chemists to relate the mass of a substance to the number of moles, which is essential for predicting reaction outcomes and determining concentrations It's one of those things that adds up..
**Q: How does the molar mass of
Answer: The molar massof Pb(NO₃)₂ is essential for converting between the mass of a sample and the number of moles that actually take part in a reaction. By knowing that one mole of Pb(NO₃)₂ weighs 331.22 g, a chemist can weigh out exactly 0.5 mol (≈ 165.61 g) to prepare a half‑molar solution, or determine that a 2.0 g sample corresponds to only 0.0060 mol, which then dictates the stoichiometric amount of another reactant required in the balanced equation Nothing fancy..
Practical Example: Preparing a Standard Lead(II) Nitrate Solution
Suppose a laboratory needs a 0.250 M Pb(NO₃)₂ solution in a 250 mL volumetric flask Small thing, real impact..
-
Calculate the required moles:
[ n = M \times V = 0.250;\text{mol·L}^{-1} \times 0.250;\text{L} = 0.0625;\text{mol} ] -
Convert moles to mass using the molar mass:
[ m = n \times M_{\text{molar}} = 0.0625;\text{mol} \times 331.22;\text{g·mol}^{-1} = 20.70;\text{g} ] -
Weigh the solid, dissolve, and dilute to the mark. Because the molar mass is known to two decimal places, the calculated mass is reliable to the nearest 0.01 g, ensuring the final concentration is accurate to within the desired tolerance (often ±0.01 M for analytical work) And that's really what it comes down to..
Linking Molar Mass to Reaction Stoichiometry
Consider the precipitation of lead(II) iodide from a solution of Pb(NO₃)₂:
[ \text{Pb(NO}_3)_2 (aq) + 2,\text{KI} (aq) \rightarrow \text{PbI}_2 (s) + 2,\text{KNO}_3 (aq) ]
If a student isolates 1.50 g of dry PbI₂ precipitate, the amount of Pb(NO₃)₂ originally present can be back‑calculated:
-
Molar mass of PbI₂:
[ M_{\text{PbI}_2}=207.2 + 2(126.9) = 461.0;\text{g·mol}^{-1} ] -
Moles of PbI₂ formed:
[ n_{\text{PbI}2}= \frac{1.50;\text{g}}{461.0;\text{g·mol}^{-1}} = 0.00325;\text{mol} ] 3. Because the stoichiometry is 1:1, the same number of moles of Pb(NO₃)₂ were consumed.
Converting back to mass of the nitrate salt:
[ m{\text{Pb(NO}_3)_2}=0.00325;\text{mol}\times 331.22;\text{g·mol}^{-1}=1.08;\text{g} ] Thus, the molar mass of Pb(NO₃)₂ enables the reverse calculation that links a measured precipitate mass to the original reagent quantity.
Safety and Handling Considerations
Lead(II) nitrate is a strong oxidizer and toxic. When working with it, the following precautions are advisable:
- Personal protective equipment: nitrile gloves, safety goggles, and a lab coat.
- Ventilation: Perform all manipulations inside a certified fume hood to avoid inhaling dust or vapors.
- Waste disposal: Collect lead‑containing waste in designated containers and follow institutional hazardous‑waste protocols.
Understanding the molar mass is not merely an academic exercise; it informs how much material must be handled, how dilute solutions should be prepared to minimize exposure, and how much waste will be generated for proper disposal No workaround needed..
Frequently Asked Follow‑Up
Q: Can the molar mass of Pb(NO₃)₂ be used to convert between mass percent and parts‑per‑million (ppm) in environmental samples?
A: Yes. By first converting a measured mass of lead (e.g., 0.004 g) to moles using the molar mass, then to volume or mass of solution, one can express the concentration in mg L⁻¹ or µg L⁻¹, which are the units commonly reported for water‑quality standards No workaround needed..
**Q: Does the presence of water of
Q: Does the presence of water of crystallization affect the molar mass used in calculations?
A: Yes. If the sample you are using is a hydrate, the water molecules are part of the solid’s mass and must be included in the molar‑mass value. Lead(II) nitrate is most commonly sold as the anhydrous salt (Pb(NO₃)₂, M ≈ 331.22 g mol⁻¹), but it can form a dihydrate (Pb(NO₃)₂·2H₂O). In that case the molar mass becomes
[ M_{\text{dihydrate}} = 331.22;\text{g·mol}^{-1} + 2(18.015;\text{g·mol}^{-1}) \approx 367.25;\text{g·mol}^{-1}. ]
When preparing a solution, using the anhydrous molar mass for a hydrated sample will give a solution that is less concentrated than intended (the actual mass of the nitrate anion per gram of solid is lower because part of the mass is water). Conversely, assuming a hydrate while working with the anhydrous form will yield a solution that is too concentrated.
To avoid error, either:
- Check the label or certificate of analysis for the specific form supplied.
- Dry the salt in a desiccator or under a gentle stream of dry air if the anhydrous form is required.
- Account for the water explicitly in your calculations if you deliberately use the hydrate.
In practice, most analytical work with lead nitrate employs the anhydrous form, and the molar mass of 331.22 g mol⁻¹ is the standard value used in stoichiometric calculations, solution preparation, and environmental reporting.
Practical Take‑aways
- Accurate molar mass is the cornerstone of quantitative work with lead(II) nitrate, whether you are preparing a standard solution, back‑calculating reagent amounts from a precipitate, or converting between mass‑based and mole‑based concentrations.
- Hydration state matters. Always verify whether your material is anhydrous or hydrated, and adjust the molar mass accordingly.
- Safety and waste considerations scale with the amount of material. Knowing the exact mass you are handling informs the selection of appropriate PPE, ventilation, and disposal containers.
Conclusion
The molar mass of lead(II) nitrate, 331.25 g mol⁻¹ for the dihydrate), is far more than a simple number on a bottle. By integrating this fundamental property into every stage of laboratory practice—from weighing the solid to disposing of the waste—chemists ensure both analytical accuracy and compliance with health‑and‑safety standards. It enables precise solution preparation, underpins stoichiometric calculations, and guides safe handling and environmental monitoring. 22 g mol⁻¹ (or 367.Understanding and applying the correct molar mass is therefore essential for anyone working with lead nitrate in teaching, research, or industrial settings.