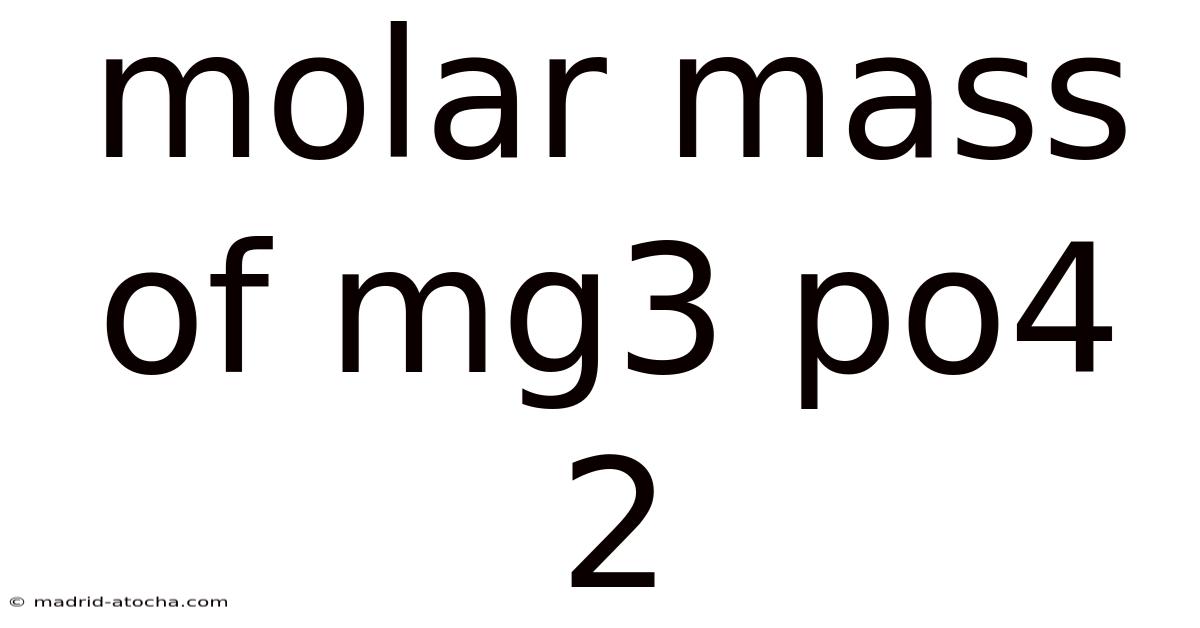Themolar mass of Mg₃(PO₄)₂ is a fundamental concept in chemistry, representing the mass of one mole of this compound. Understanding this value is crucial for numerous calculations, from determining the composition of compounds to quantifying reactions. This article provides a full breakdown to calculating the molar mass of magnesium phosphate, breaking down the process into clear, manageable steps and explaining the underlying science.
You'll probably want to bookmark this section Small thing, real impact..
Introduction
Molar mass, often denoted as M, is the mass of one mole of a substance, expressed in grams per mole (g/mol). A mole is a unit representing 6.That's why 022 × 10²³ entities (Avogadro's number), allowing chemists to work with manageable quantities of atoms and molecules. Calculating the molar mass of a compound involves summing the atomic masses of all atoms present in its chemical formula. For ionic compounds like Mg₃(PO₄)₂, this process is straightforward once you understand the formula and the atomic masses involved Not complicated — just consistent..
Steps to Calculate the Molar Mass of Mg₃(PO₄)₂
Calculating the molar mass of Mg₃(PO₄)₂ follows a simple, systematic approach:
- Identify the Formula: The formula is Mg₃(PO₄)₂. This indicates one magnesium (Mg) atom is surrounded by two phosphate (PO₄) groups. The subscript "3" applies to the entire (PO₄)₂ unit.
- Break Down the Formula: Identify the elements and their quantities:
- Magnesium (Mg): Subscript 3 (Mg₃)
- Phosphorus (P): Subscript 2 (PO₄)₂ → 2 P atoms
- Oxygen (O): Subscript 4 (PO₄)₂ → 4 O atoms per phosphate group, multiplied by 2 groups → 8 O atoms
- Recall Atomic Masses: Use the standard atomic masses from the periodic table:
- Magnesium (Mg): 24.305 g/mol
- Phosphorus (P): 30.974 g/mol
- Oxygen (O): 15.999 g/mol
- Calculate the Mass Contribution of Each Element:
- Mass of Mg: 3 atoms × 24.305 g/mol = 72.915 g/mol
- Mass of P: 2 atoms × 30.974 g/mol = 61.948 g/mol
- Mass of O: 8 atoms × 15.999 g/mol = 127.992 g/mol
- Sum the Masses: Add the contributions from all elements to find the total molar mass:
- Molar Mass of Mg₃(PO₄)₂ = Mass of Mg + Mass of P + Mass of O
- = 72.915 g/mol + 61.948 g/mol + 127.992 g/mol
- = 262.855 g/mol
Because of this, the molar mass of Mg₃(PO₄)₂ is 262.855 grams per mole Not complicated — just consistent..
Scientific Explanation
The molar mass calculation relies on two fundamental principles: the definition of a mole and the conservation of mass. Even so, a mole of Mg₃(PO₄)₂ contains exactly 3 moles of Mg atoms, 2 moles of P atoms, and 8 moles of O atoms. The molar mass is simply the sum of the masses of these constituent atoms.
Easier said than done, but still worth knowing.
- Atomic Mass: The atomic mass of an element listed on the periodic table is the weighted average mass of its naturally occurring isotopes, expressed in atomic mass units (amu). For calculation purposes, we use these values in grams per mole (g/mol). Take this: the atomic mass of Mg is 24.305 g/mol, meaning one mole of Mg atoms weighs 24.305 grams.
- Formula Weight vs. Molecular Weight: For ionic compounds like Mg₃(PO₄)₂, which do not form discrete molecules, we refer to the formula weight (FW). The formula weight is calculated identically to the molar mass (g/mol). It represents the mass of one formula unit of the compound. In this case, the formula unit is Mg₃(PO₄)₂. While molecular weight applies to covalent molecular compounds, formula weight is the correct term for ionic compounds.
FAQ
- Why is the subscript "3" outside the parentheses in Mg₃(PO₄)₂?
- The subscript "3" applies to the entire phosphate group (PO₄)₂. This means there are 3 magnesium atoms for every 2 phosphate groups. The parentheses group the atoms of the PO₄ unit together, indicating they are bonded as a single entity. Without the parentheses, Mg3PO4 would imply 3 Mg, 1 P, and 4 O atoms, which is incorrect for this compound.
- What's the difference between molar mass and molecular weight?
- For covalent molecular compounds (like H₂O), the terms are often used interchangeably. Still, for ionic compounds (like Mg₃(PO₄)₂), we use formula weight because the compound exists as a lattice of ions, not discrete molecules. The calculation method is the same, but the terminology differs.
- Why do we use the atomic masses from the periodic table?
- The atomic masses listed on the periodic table are the standard reference values used in chemistry. They account for the average mass of all naturally occurring isotopes of an element. These values are universally accepted for calculating molar masses.
- How is the molar mass of Mg₃(PO₄)₂ used in practice?
- It's essential for:
- Calculating the mass of a specific number of moles (mass = moles × molar mass).
- Determining the empirical and molecular formulas of compounds.
- Balancing chemical equations quantitatively.
- Calculating concentrations (molarity) of solutions.
- Performing stoichiometric calculations in reactions involving magnesium phosphate.
- It's essential for:
- Can I use rounded atomic masses for a quick estimate?
- Yes, for rough estimates, you can use rounded values (e.g., Mg = 24, P = 31, O = 16). Using Mg = 24, P = 31, O = 16:
- Mg: 3 × 24 = 72
- P: 2 × 31 = 62
- O: 8 × 16 = 128
- Total ≈ 72 + 62 + 128 = 262 g/mol
- This gives a close approximation (262 vs. 262.855) suitable for many calculations. Still, for precise work, always use the
- Yes, for rough estimates, you can use rounded values (e.g., Mg = 24, P = 31, O = 16). Using Mg = 24, P = 31, O = 16:
unrounded atomic masses provided by the periodic table.
Conclusion
Understanding the distinction between formula weight and molecular weight is fundamental to accurately working with chemical compounds. The ability to correctly calculate and interpret these values empowers us to understand and predict chemical behavior, paving the way for advancements in various scientific fields, from materials science to medicine. Even so, this knowledge is vital for a wide range of chemical calculations, from determining reaction yields to preparing solutions. Practically speaking, while both concepts relate to mass, their application hinges on the compound's structure. Practically speaking, formula weight provides a precise measure of the mass of a formula unit for ionic compounds, reflecting their ionic nature. By mastering these concepts and utilizing the correct terminology, students and professionals alike can confidently work through the quantitative aspects of chemistry and perform calculations with accuracy and precision. When all is said and done, a firm grasp of formula weight and molecular weight is a cornerstone of chemical literacy.
Counterintuitive, but true.
Further Considerations and Applications
Beyond the core calculations, understanding formula weight and molecular weight unlocks a deeper appreciation for chemical bonding and stoichiometry. Consider how these values relate to the strength of chemical bonds. Consider this: for example, compounds with higher formula weights often exhibit greater stability due to the increased mass of the molecules. This concept is crucial in predicting the properties of different chemical substances.
To build on this, the concept of molar mass is directly linked to Avogadro's number, a fundamental constant in chemistry. Now, molar mass allows us to relate the mass of a substance to the number of particles (atoms, molecules, ions) present, forming the basis for understanding chemical reactions at a microscopic level. This connection is essential for interpreting experimental data and developing theoretical models.
In practical applications, the understanding of formula weight and molar mass is indispensable in various fields. In pharmaceutical chemistry, accurate molar mass calculations are crucial for drug formulation and dosage calculations. In real terms, in environmental science, they are essential for determining the concentrations of pollutants in water and air. In materials science, they play a vital role in designing and synthesizing new materials with specific properties. Beyond that, in analytical chemistry, these values are used in quantitative analysis techniques to determine the composition of unknown samples Worth keeping that in mind..
Which means, a thorough understanding of formula weight and molar mass isn't just a theoretical exercise; it's a practical skill that underpins countless aspects of modern science and technology. Continued study and application of these concepts will undoubtedly contribute to further advancements in our understanding of the chemical world The details matter here..