In Which Reaction Does The Oxidation Number Of Oxygen Increase
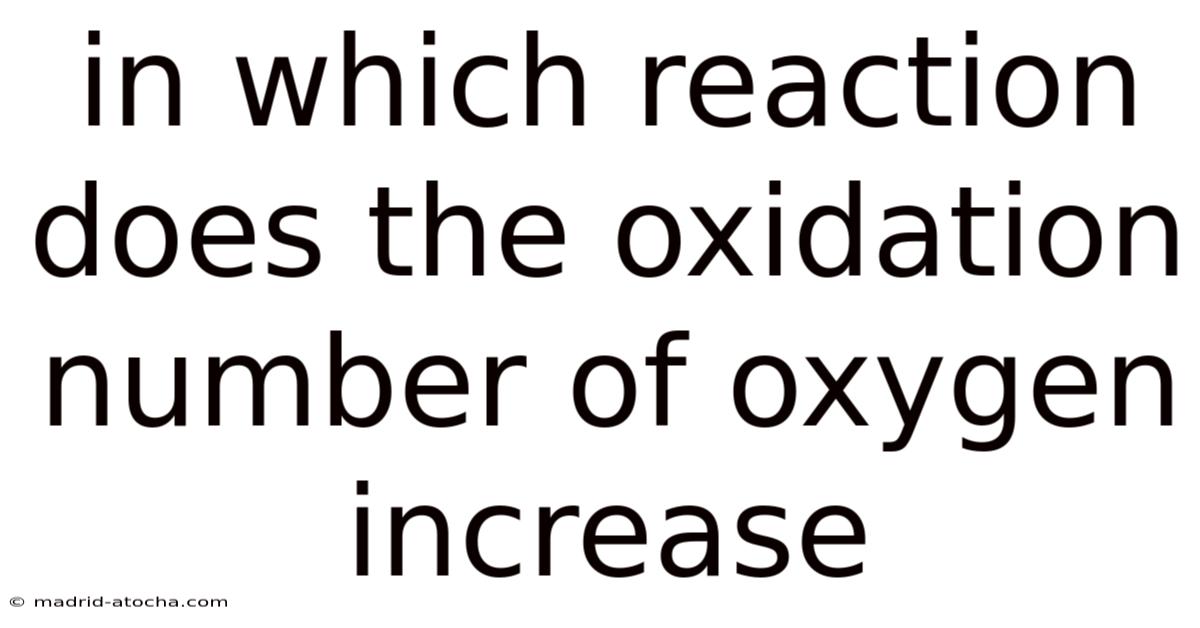
In the realm of redox chemistry, the oxidation number of oxygen almost invariably decreases, reflecting its high electronegativity and its typical role as an oxidizing agent. However, specific and fascinating reactions exist where oxygen’s oxidation state actually increases, moving from its common -2 value toward zero or even positive integers. These exceptions are not merely academic curiosities; they reveal fundamental principles of chemical bonding and reactivity, particularly when oxygen encounters elements more electronegative than itself or participates in unique molecular structures. Understanding these reactions provides a deeper, more nuanced grasp of oxidation-reduction principles beyond the standard textbook examples.
The Usual Role of Oxygen: A Powerful Oxidizer
To appreciate the exceptions, one must first understand the rule. Oxygen is the second most electronegative element (3.44 on the Pauling scale), surpassed only by fluorine (3.98). This powerful electron-withdrawing tendency means that in virtually all its compounds—with the notable exception of those with fluorine—oxygen adopts an oxidation number of -2. In water (H₂O), carbon dioxide (CO₂), and metal oxides like iron(III) oxide (Fe₂O₃), oxygen’s oxidation state is -2. In these compounds, oxygen is reduced (gains electrons) during its formation from elemental oxygen (O₂, oxidation number 0). Consequently, in most reactions, oxygen acts as the oxidizing agent, causing other substances to be oxidized while it is itself reduced (its oxidation number decreases).
Key Reactions Where Oxygen’s Oxidation Number Increases
1. Reaction with Fluorine: The Primary Exception
The only element capable of consistently oxidizing oxygen is fluorine. Due to fluorine’s superior electronegativity, it forces oxygen to adopt a positive oxidation state in their binary compound, oxygen difluoride (OF₂).
- Chemical Reaction: O₂ + 2F₂ → 2OF₂
- **Oxidation Number Change
...Analysis: In this reaction, the oxidation state of fluorine increases from 0 to -1, while the oxidation state of oxygen increases from -2 to +2. This demonstrates that fluorine's extreme electronegativity can override the typical behavior of oxygen in forming stable compounds. This reaction is highly energetic and often occurs under extreme conditions, highlighting the exceptional nature of this oxidation state change.
2. Oxygen in Peroxides: A Complex Case
Peroxides, such as hydrogen peroxide (H₂O₂) and various metal peroxides (e.g., potassium peroxide, K₂O₂), present a more nuanced scenario. While the overall oxidation state of the peroxide ion (O₂⁻) is -1, the oxygen atoms within the peroxide molecule can exhibit a range of oxidation states, including +1 and even +2, depending on the specific peroxide and the reaction conditions. This is due to the delocalization of electrons within the peroxide structure and the influence of the surrounding atoms.
- Hydrogen Peroxide (H₂O₂): The oxygen atoms in H₂O₂ are typically considered to have an oxidation state of +1, though the exact value can be debated due to the molecule's bent geometry and the presence of lone pairs. This is a deviation from the usual -2 rule, but not a complete reversal.
- Metal Peroxides: The oxidation state of oxygen in metal peroxides (like K₂O₂) is often +2, but this is not always the case and can vary depending on the metal and the specific peroxide.
3. Oxygen in Certain Organic Compounds: A Less Common Occurrence
While less frequent, oxygen can sometimes exhibit a positive oxidation state in certain organic compounds. This typically occurs in highly reactive species or in specific reaction pathways. For example, in some complex organic reactions involving radical intermediates, oxygen can be transiently oxidized to form positive oxidation states. These cases are often highly context-dependent and require careful consideration of the overall reaction mechanism.
Conclusion:
The seemingly paradoxical increase in oxygen’s oxidation number, as seen in reactions with fluorine and certain peroxides, underscores the multifaceted nature of chemical behavior. While oxygen’s propensity for a -2 oxidation state is firmly established, its ability to adopt positive oxidation states reveals the intricate interplay of electronegativity, bonding, and reactivity. These exceptions are not anomalies but rather valuable illustrations of fundamental chemical principles. They highlight how the behavior of elements can be significantly influenced by the surrounding environment and the specific chemical context. By studying these unusual reactions, chemists gain a deeper understanding of the underlying mechanisms of oxidation-reduction processes, moving beyond rote memorization of standard examples to a more nuanced and comprehensive appreciation of chemical reactivity. This expanded understanding is crucial for developing new chemical technologies and for interpreting complex chemical phenomena observed in various fields, from materials science to biochemistry.
Latest Posts
Latest Posts
-
When A Food Recall Occurs The Operation Must
Mar 20, 2026
-
A Person Loses 2 70 Lb In Two Weeks
Mar 20, 2026
-
Excel 2021 In Practice Ch 4 Guided Project 4 3
Mar 20, 2026
-
Bark In The Park Teas Passage
Mar 20, 2026
-
Physical Geography Lab Manual 12th Edition Answers
Mar 20, 2026