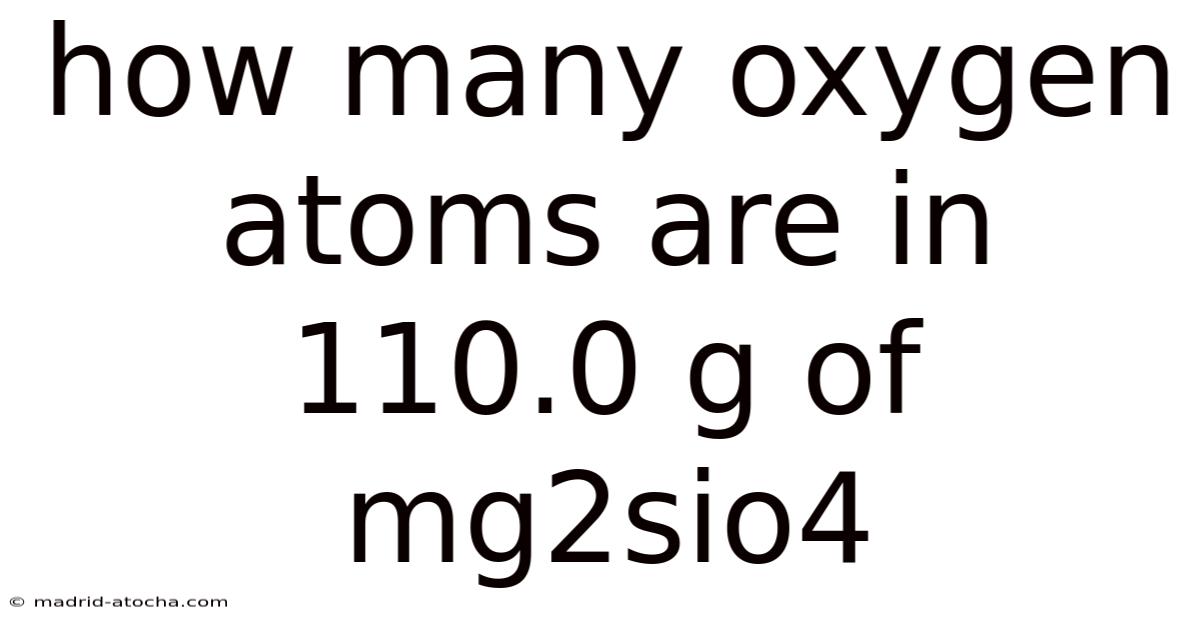How Many Oxygen Atoms Are in 110.0 g of Mg₂SiO₄?
If you’ve ever wondered how many oxygen atoms are packed into a specific mass of a compound like magnesium silicate (Mg₂SiO₄), you’re not alone. This question bridges chemistry, stoichiometry, and real-world applications, from material science to environmental studies. Let’s break it down step by step to uncover the answer Easy to understand, harder to ignore..
Step-by-Step Calculation: From Grams to Atoms
To determine the number of oxygen atoms in 110.0 g of Mg₂SiO₄, we follow a systematic approach rooted in chemical principles.
1. Understand the Molecular Formula
Magnesium silicate (Mg₂SiO₄) consists of:
- 2 magnesium (Mg) atoms
- 1 silicon (Si) atom
- 4 oxygen (O) atoms
This means every molecule of Mg₂SiO₄ contains 4 oxygen atoms Worth knowing..
2. Calculate the Molar Mass of Mg₂SiO₄
The molar mass is the sum of the atomic masses of all atoms in the compound:
- Mg: 24.305 g/mol × 2 = 48.61 g/mol
- Si: 28.085 g/mol × 1 = 28.085 g/mol
- O: 16.00 g/mol × 4 = 64.00 g/mol
Total molar mass = 48.61 + 28.085 + 64.00 = 140.695 g/mol
3. Convert Grams to Moles of Mg₂SiO₄
Use the formula:
$ \text{Moles} = \frac{\text{Mass (g)}}{\text{Molar Mass (g/mol)}} $
Plugging in the values:
$ \text{Moles of Mg₂SiO₄} = \frac{110.0 , \text{g}}{140.695 , \text{g/mol}} \approx 0.7816 ,