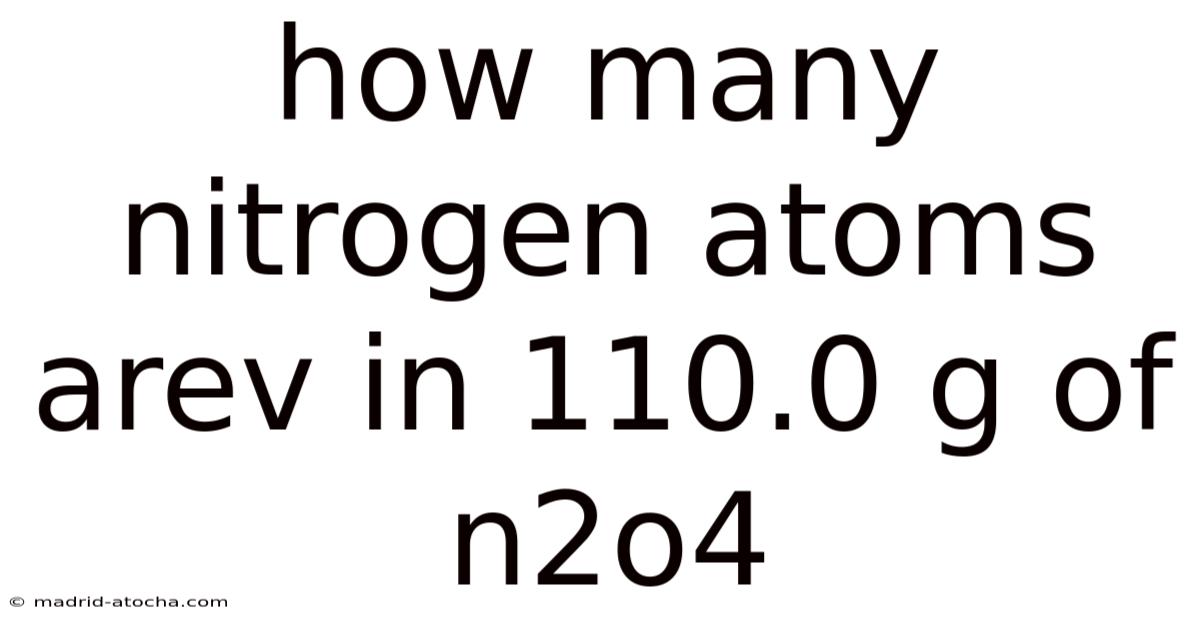How Many Nitrogen Atoms Are in 110.0 g of N₂O₄? – A Step‑by‑Step Guide
When a chemistry student sees the question “how many nitrogen atoms are in 110.” the first instinct is often to reach for a calculator and start plugging numbers. Even so, 0 g of N₂O₄? Yet the problem is an excellent opportunity to review fundamental concepts—molar mass, Avogadro’s number, and stoichiometric conversion—while also sharpening the ability to translate a mass into an exact count of atoms. This article walks you through every stage of the calculation, explains the scientific reasoning behind each step, and provides useful tips for similar quantitative problems And that's really what it comes down to..
Introduction: From Grams to Atoms
The core of the problem is a classic mass‑to‑quantity conversion. We are given a mass (110.0 g) of a molecular compound, dinitrogen tetroxide (N₂O₄), and we must determine the total number of nitrogen atoms contained in that sample.
This changes depending on context. Keep that in mind Small thing, real impact..
- Molar mass of N₂O₄ – the mass of one mole of the compound.
- Avogadro’s constant (Nₐ = 6.022 × 10²³) – the number of entities (atoms, molecules, ions) per mole.
- Stoichiometric factor – each N₂O₄ molecule carries two nitrogen atoms.
By chaining these three relationships together, we can move from grams → moles of N₂O₄ → moles of nitrogen atoms → number of nitrogen atoms.
Step 1: Calculate the Molar Mass of N₂O₄
The molar mass is obtained by summing the atomic masses of all atoms in the formula.
| Element | Symbol | Atomic mass (g mol⁻¹) | Quantity in N₂O₄ |
|---|---|---|---|
| Nitrogen | N | 14.01 | 2 |
| Oxygen | O | 16.00 | 4 |
[ \text{Molar mass of N₂O₄}= (2 \times 14.Think about it: 01) + (4 \times 16. 00) = 28.02 + 64.00 = 92 Easy to understand, harder to ignore..
Note: The value 92.02 g mol⁻¹ is typically rounded to 92.0 g mol⁻¹ when using three significant figures, matching the three‑significant‑figure precision of the given mass (110.0 g) Less friction, more output..
Step 2: Convert the Given Mass to Moles of N₂O₄
Using the definition of the mole:
[ \text{moles of N₂O₄}= \frac{\text{mass (g)}}{\text{molar mass (g mol⁻¹)}} ]
[ \text{moles of N₂O₄}= \frac{110.0\ \text{g}}{92.0\ \text{g mol}^{-1}} = 1.
Rounded to three significant figures, 1.20 mol of N₂O₄ are present Small thing, real impact..
Step 3: Determine the Moles of Nitrogen Atoms
Each N₂O₄ molecule contains two nitrogen atoms. That's why, the mole ratio N (atoms) : N₂O₄ (molecules) is 2 : 1.
[ \text{moles of N atoms}= 2 \times \text{moles of N₂O₄}= 2 \times 1.20\ \text{mol}= 2.40\ \text{mol} ]
Step 4: Convert Moles of Nitrogen Atoms to Number of Atoms
Finally, apply Avogadro’s constant:
[ \text{Number of N atoms}= (2.40\ \text{mol}) \times (6.022 \times 10^{23}\ \text{atoms mol}^{-1}) ]
[ \text{Number of N atoms}= 1.45 \times 10^{24}\ \text{atoms} ]
Rounded to three significant figures, 1.Consider this: 45 × 10²⁴ nitrogen atoms are contained in 110. 0 g of N₂O₄ It's one of those things that adds up..
Scientific Explanation: Why the Method Works
The Mole Concept
The mole bridges the macroscopic world (grams you can weigh) and the microscopic world (atoms you cannot see). Think about it: 022 × 10²³ entities**, chemists can count particles indirectly. Consider this: by defining **one mole as exactly 6. The molar mass, expressed in grams per mole, tells us how much of a substance corresponds to that count.
Stoichiometry and Molecular Formula
A chemical formula is more than a label; it encodes the ratio of atoms inside each molecule. For N₂O₄, the subscript “₂” tells us there are two nitrogen atoms per molecule, while “₄” indicates four oxygen atoms. When we multiply the number of moles of molecules by this subscript, we obtain the moles of the specific element Worth keeping that in mind..
Significant Figures and Precision
The given mass (110.Now, 01 g mol⁻¹ for N, 16. In practice, 00 g mol⁻¹ for O) limit the final answer to three significant figures. Still, 0 g) has four significant figures, but the atomic masses used (14. Maintaining consistent precision throughout the calculation prevents false accuracy.
Frequently Asked Questions (FAQ)
1. What if the mass were given with more or fewer significant figures?
Adjust the final answer to match the least‑precise input. Here's one way to look at it: 110 g (three significant figures) would still yield 1.45 × 10²⁴ N atoms, but you would report it as 1.45 × 10²⁴ rather than 1.450 × 10²⁴.
2. Can I use the atomic weight of nitrogen as 14.007 instead of 14.01?
Yes. Using more precise atomic masses will slightly change the intermediate molar mass (92.011 g mol⁻¹) and the final count, but the difference will be within the uncertainty dictated by the given mass.
3. Why do we multiply by 2 in Step 3?
The factor 2 reflects the stoichiometric coefficient of nitrogen in the molecular formula. Each N₂O₄ molecule contributes two nitrogen atoms, so the number of nitrogen moles is twice the number of N₂O₄ moles.
4. Is it necessary to convert to moles of nitrogen atoms before applying Avogadro’s number?
Yes. Avogadro’s constant relates moles of entities to individual entities. Skipping the mole‑to‑atom conversion would give the number of N₂O₄ molecules, not nitrogen atoms.
5. How would the calculation change for a different compound, such as NH₃?
The steps remain identical; only the molar mass and the stoichiometric coefficient for nitrogen would differ (NH₃ contains one nitrogen atom per molecule) That's the part that actually makes a difference..
Practical Tips for Similar Problems
| Tip | Reason |
|---|---|
| Write the formula clearly | Prevents forgetting subscripts (e. |
| List atomic masses before calculating | Saves time and reduces arithmetic errors. NO₂). Here's the thing — |
| Keep track of units | Cancel g with g mol⁻¹ to obtain mol; then mol × atoms mol⁻¹ = atoms. , N₂O₄ vs. But |
| Use scientific notation for large numbers | Makes the final answer readable (10²⁴ instead of 1,450,000,000,000,000,000,000,000). g. |
| Check significant figures at the end | Guarantees the answer reflects the precision of the data. |
Conclusion: From 110 g to 1.45 × 10²⁴ Atoms
The question “how many nitrogen atoms are in 110.0 g of N₂O₄?” may appear intimidating at first glance, but it is a straightforward application of the mole concept, molar mass calculation, and Avogadro’s constant.
- Determining the molar mass of N₂O₄ (≈ 92.0 g mol⁻¹),
- Converting the given mass to moles of N₂O₄ (≈ 1.20 mol),
- Multiplying by the stoichiometric factor of nitrogen (2) to get moles of N atoms (≈ 2.40 mol), and
- Using Avogadro’s number to obtain the absolute count (≈ 1.45 × 10²⁴ atoms),
we arrive at a precise, scientifically sound answer. Mastering this sequence not only solves the immediate problem but also builds a dependable framework for tackling any mass‑to‑atom conversion in chemistry. Whether you are preparing for an exam, writing a lab report, or simply satisfying curiosity, the method outlined here will serve as a reliable, repeatable tool in your analytical toolkit.