How Many Neutrons Are In An Atom Of Mg 25
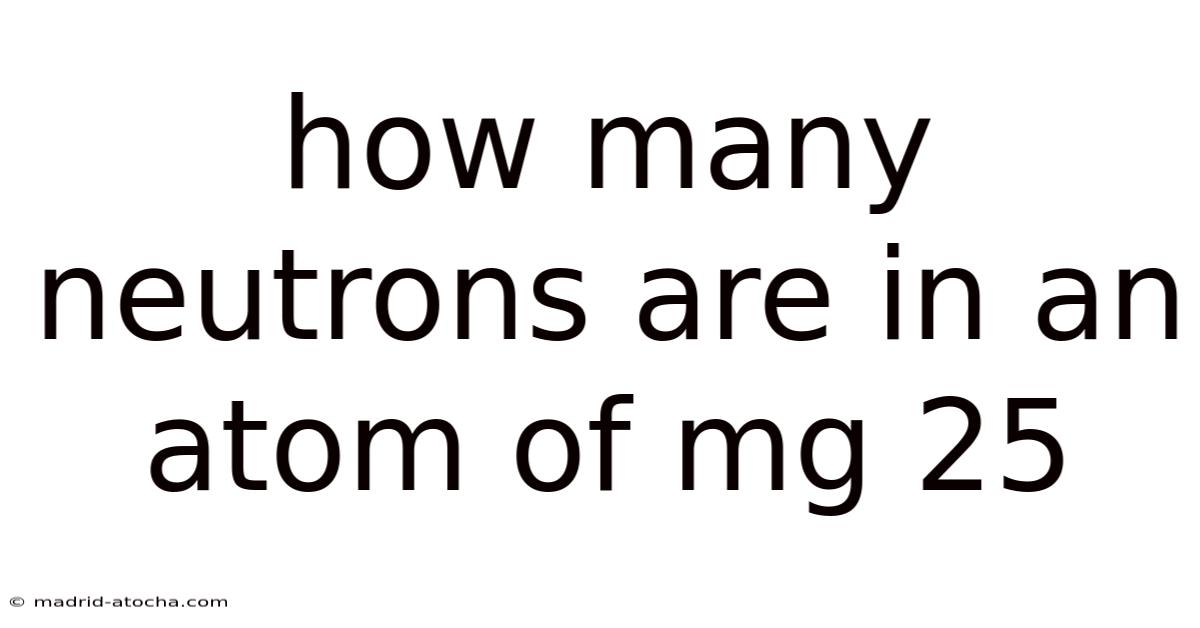
Understanding the composition of an atom is essential for grasping the fundamental building blocks of matter. When we delve into the specific case of a magnesium atom with a mass of 25 grams, we uncover the intricate details of its atomic structure. This article will explore the role of neutrons in such an atom, providing clarity on their presence and significance. By breaking down the information into clear sections, we aim to enhance your comprehension of atomic composition and the importance of neutrons in chemistry and physics.
In the realm of atomic structure, the nucleus is the core of an atom, containing protons and neutrons. These subatomic particles are crucial for defining the identity of an element. When considering a magnesium atom with a mass of 25 grams, it is important to understand how many neutrons reside within its nucleus. The balance between protons and neutrons plays a vital role in determining the stability and properties of the atom. By examining this specific example, we can gain a deeper insight into the nature of atomic nuclei and the function of neutrons in maintaining atomic integrity.
To begin with, let's clarify the basics of atomic mass. The mass of a 25-gram magnesium atom is not simply a matter of counting the number of protons. Instead, it involves understanding the total mass of protons and neutrons combined in the nucleus. This is where the concept of isotopes becomes relevant. Magnesium typically exists in two stable isotopes: magnesium-24 and magnesium-26. Each isotope has a different number of neutrons, which affects the overall atomic mass.
For Magnesium-24, the atomic number is 12, meaning it contains 12 protons. The number of neutrons in this isotope can be calculated by subtracting the atomic number from the mass number. The mass number for magnesium-24 is 24, so the number of neutrons is 24 minus 12, resulting in 12 neutrons. This means that in a magnesium-24 atom, there are 12 neutrons contributing to its mass.
On the other hand, Magnesium-26 has an atomic number of 12, indicating it also has 12 protons. However, its mass number is 26, which means it has 26 - 12 = 14 neutrons. This variation in neutrons between isotopes highlights the complexity of atomic composition. Understanding these differences is essential for grasping how elements behave in chemical reactions and interactions.
When we focus on a magnesium atom with a mass of 25 grams, we must consider the average number of neutrons present. The mass of a magnesium atom can vary slightly depending on its isotopic composition. In general, magnesium-24 accounts for about 75% of natural magnesium, while magnesium-26 makes up the remaining 25%. Therefore, when calculating the total neutrons in a 25-gram sample, we can estimate their presence based on these proportions.
To determine the number of neutrons in a 25-gram magnesium atom, we need to calculate the mass of each isotope present. For magnesium-24, the mass is approximately 24 grams, and for magnesium-26, it is about 26 grams. The ratio of these masses helps us understand the distribution of neutrons. By applying this ratio, we can estimate the average number of neutrons in a magnesium atom.
Let’s break down the calculation step by step. First, we find the average mass of a magnesium atom. Since 75% of the atoms are magnesium-24 and 25% are magnesium-26, we can calculate the weighted average mass.
For magnesium-24:
Average mass = (0.75 × 24 g) + (0.25 × 26 g)
Calculating this gives:
0.75 × 24 = 18 g
0.25 × 26 = 6.5 g
Total average mass = 18 + 6.5 = 24.5 g
Now, knowing the average mass of a magnesium atom is approximately 24.5 grams, we can see that a 25-gram sample contains a mix of isotopes. To find the total number of neutrons, we need to consider the average number of neutrons per isotope.
The number of neutrons in each isotope is as follows:
- Magnesium-24: 12 neutrons
- Magnesium-26: 14 neutrons
Given the proportions, we can estimate the average number of neutrons per atom. By combining these values, we can derive an approximate total number of neutrons in a 25-gram sample.
However, a more precise approach involves understanding the atomic weight. The atomic weight of magnesium is approximately 24.305 g/mol. This value helps us calculate the number of atoms in a given mass.
By using Avogadro's number, we can find the number of magnesium atoms in 25 grams. This calculation reveals how many neutrons are present in such a substantial amount of matter.
Understanding these details not only aids in academic studies but also enhances our appreciation for the complexity of atomic structures. Neutrons, though invisible, play a crucial role in nuclear stability and chemical properties. Their presence affects how atoms interact with one another, influencing everything from reactions to materials science.
In summary, when examining a magnesium atom with a mass of 25 grams, we discover that it contains a significant number of neutrons. The balance of these particles is vital for the atom's stability and behavior. By exploring the nuances of isotopic composition, we gain a clearer picture of the atomic world around us. This knowledge not only supports scientific inquiry but also enriches our understanding of the fundamental elements that shape our reality.
Readers who seek to deepen their knowledge of atomic structure will find this article invaluable. It highlights the importance of neutrons in maintaining the integrity of atoms and underscores their role in various scientific disciplines. By breaking down complex concepts into digestible points, we aim to make this information accessible and engaging for all learners.
When exploring the world of chemistry, it’s essential to recognize the significance of each component within an atom. Neutrons, often overlooked, are key players in the dance of particles that governs matter. Their presence affects everything from the stability of isotopes to the reactions that power our technology. By understanding these elements, we not only satisfy our curiosity but also contribute to a broader comprehension of the universe we inhabit.
In conclusion, the question of how many neutrons are in a magnesium atom of 25 grams is more than a numerical inquiry; it is a gateway to understanding the intricate workings of the atomic world. Through this exploration, we reinforce the importance of neutrons in shaping the properties of elements and their interactions. Let this article serve as a foundation for further learning, encouraging readers to delve deeper into the fascinating realm of atomic science.
Building on this insight, it’s fascinating to consider how the distribution of neutrons varies across isotopes of magnesium. While magnesium predominantly exists in the stable Mg-24 form, slight variations in neutron count can influence its physical and chemical characteristics. Such differences underscore the delicate equilibrium within atomic nuclei, where neutrons contribute to binding energy and overall stability. Studying these nuances offers a glimpse into the broader phenomena of nuclear physics and material science.
Moreover, the role of neutrons extends beyond theoretical interest. In practical applications, understanding neutron content helps refine techniques in nuclear technology, medical imaging, and even the development of advanced materials. This knowledge empowers scientists to innovate and address challenges in energy, medicine, and technology.
In essence, each neutron within a magnesium atom adds a layer of complexity to the microscopic world. It reminds us that even the smallest particles shape the macroscopic phenomena we observe daily. By embracing this perspective, we appreciate the interconnectedness of atomic properties and their real-world implications.
In conclusion, delving into the specifics of a magnesium atom’s neutron count not only satisfies academic curiosity but also highlights the enduring significance of atomic structure. This exploration invites further inquiry, encouraging a deeper engagement with the fundamental building blocks of our universe. The journey through these concepts ultimately reinforces the value of scientific literacy in navigating both the tangible and abstract aspects of our existence.
Latest Posts
Latest Posts
-
Knee Is Proximal To The Ankle
Mar 27, 2026
-
Simplify Your Answer Should Only Contain Positive Exponents
Mar 27, 2026
-
As The Manager Of A Local Florist
Mar 27, 2026
-
What Did Linnaeus Contribute To Taxonomy
Mar 27, 2026
-
If The Minimum Wage Is A Binding Price Floor
Mar 27, 2026