How Many Moles Of Calcium Are In 1.00 G
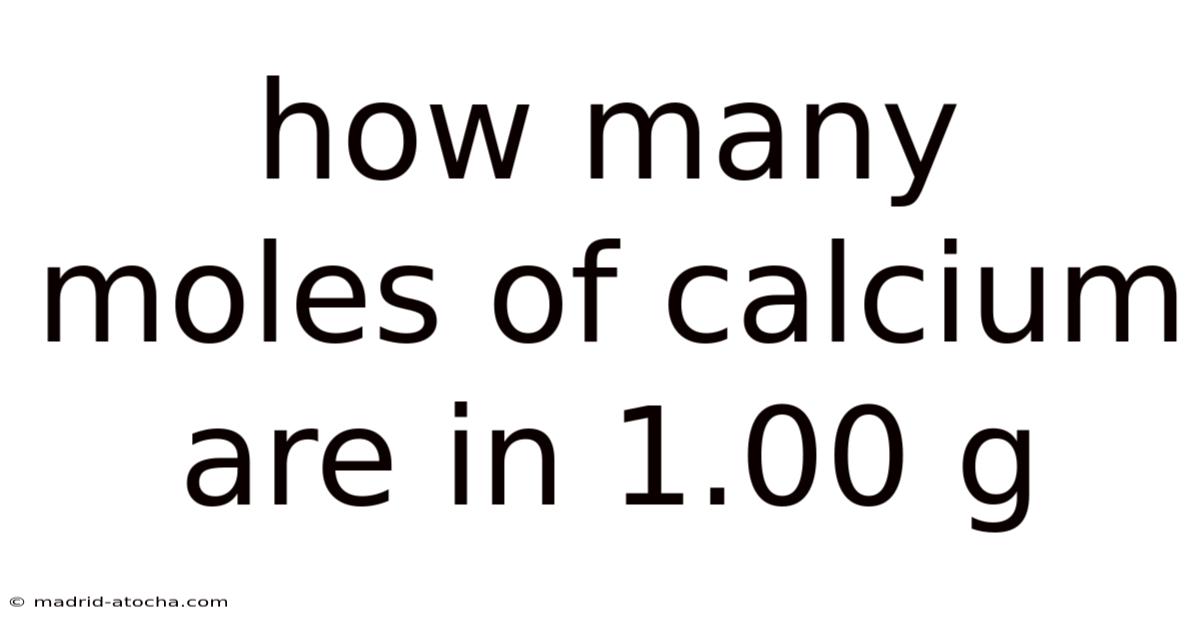
How Many Moles of Calcium Are in 1.00 g?
Understanding the relationship between mass and moles is a cornerstone of chemistry, enabling scientists to quantify substances at the atomic level. When asked, “How many moles of calcium are in 1.00 g?”, the answer lies in the concept of molar mass—a bridge between the macroscopic world of grams and the microscopic world of atoms. This article explores the calculation, its scientific basis, and its practical applications in chemistry.
Understanding Moles and Molar Mass
A mole is a unit in chemistry that represents a specific number of particles, such as atoms, molecules, or ions. One mole contains Avogadro’s number of particles, approximately 6.022 × 10²³. This number allows chemists to count atoms indirectly by measuring mass.
The molar mass of an element is the mass of one mole of its atoms, expressed in grams per mole (g/mol). It is calculated by summing the atomic weights of all atoms in a molecule, as listed on the periodic table. For calcium (Ca), the atomic weight is 40.08 g/mol, meaning one mole of calcium atoms weighs 40.08 grams.
Step-by-Step Calculation: Moles of Calcium in 1.00 g
To determine the number of moles in 1.00 g of calcium, follow these steps:
-
Identify the given mass:
The mass of calcium is 1.00 g. -
Find the molar mass of calcium:
From the periodic table, calcium’s atomic weight is 40.08 g/mol. -
Apply the formula:
$ \text{Moles} = \frac{\text{Mass (g)}}{\text{Molar Mass (g/mol)}} $
Substituting the values:
$ \text{Moles of Ca} = \frac{1.00\ \text{g}}{40.08\ \text{g/mol}} \approx 0.02495\ \text{mol} $ -
Round to significant figures:
Since the given mass (1.00 g) has three significant figures, the result is rounded to 0.0250 mol.
Scientific Explanation: Why Moles Matter
The mole concept simplifies chemical calculations by linking mass to the number of atoms. For example, 1.00 g of calcium contains 0.0250 moles, which corresponds to 1.50 × 10²² atoms (calculated by multiplying moles by Avogadro’s number: $0.0250\ \text{mol} \times 6.022 \times 10^{23}\ \text{atoms/mol}$). This relationship is critical in stoichiometry, where precise measurements ensure reactions proceed efficiently.
Molar mass also reflects the periodic table’s structure. Calcium’s atomic weight (40.08) arises from its 20 protons, 20 neutrons, and 20 electrons in a neutral atom. Variations in isotopic composition (e.g., calcium-40, calcium-42) slightly alter the average atomic weight, but the value 40.08 g/mol is widely accepted for practical calculations.
Real-World Applications of Molar Mass
-
Industrial Chemistry:
In manufacturing, knowing the moles of calcium in a sample ensures accurate dosing in processes like cement production or water softening. -
Pharmaceuticals:
Calcium supplements are formulated using molar calculations to deliver the correct dosage. For instance, a 1.00 g tablet contains ~0.025 moles of calcium, aligning with recommended daily intakes. -
Environmental Science:
Soil scientists use molar mass to assess calcium levels in agricultural land, optimizing crop growth.
FAQ: Common Questions About Moles and Calcium
Q: Why is the molar mass of calcium 40.08 g/mol?
A: The molar mass is derived from the average atomic weight of calcium’s isotopes, weighted by their natural abundance.
Q: Can I use grams directly in chemical equations?
A: No. Chemical equations require moles to represent the ratio of reactants and products. Grams must first be converted to moles.
Q: What if the mass is not 1.00 g?
A: The formula remains the same. For example, 2.00 g of calcium would yield $ \frac{2.00}{40.08} \approx 0.0500\ \text{mol} $.
Q: How does this relate to Avogadro’s number?
A: Moles provide a bridge between
Q: How does this relate to Avogadro’s number? A: As demonstrated, moles are directly proportional to Avogadro’s number (6.022 x 10²³ entities per mole). This allows us to convert between the macroscopic world of grams and the microscopic world of atoms and molecules, providing a crucial link for understanding and predicting chemical reactions.
Q: What are some potential sources of error when determining molar mass? A: While 40.08 g/mol is a standard value, slight variations can occur due to the presence of calcium isotopes, primarily calcium-40 and calcium-42. The abundance of these isotopes varies slightly depending on the source material. Furthermore, experimental determination of atomic weights relies on precise mass spectrometry, and minor instrument errors can introduce small discrepancies. However, for most practical applications, the accepted value of 40.08 g/mol is sufficiently accurate.
Q: Beyond calcium, how does the mole concept apply to other elements and compounds? A: The mole concept is universally applicable to all substances. The molar mass of any element is found on the periodic table and represents the mass of one mole of that element in grams. For compounds, the molar mass is calculated by summing the molar masses of all the atoms present in the chemical formula. For example, water (H₂O) has a molar mass of (2 * 1.008) + 16.00 = 18.016 g/mol.
Conclusion
The mole, a deceptively simple concept, is a cornerstone of modern chemistry. From accurately calculating reaction yields to formulating pharmaceuticals and assessing environmental conditions, understanding and applying the mole concept is essential for chemists and scientists across numerous disciplines. By linking mass to the number of particles, the mole provides a powerful tool for bridging the gap between the macroscopic and microscopic worlds, enabling precise and predictable chemical behavior. The seemingly small number – 6.022 x 10²³ – represents a fundamental relationship that underpins our understanding of matter and its interactions.
Latest Posts
Latest Posts
-
The Right Communication Channel To Use In Imc Is
Mar 28, 2026
-
Which Periodic Reinvestigation Is Required For Continued Secret Clearance
Mar 28, 2026
-
The Drawing Shows A Square Each Side Of Which
Mar 28, 2026
-
When The Wash Sale Rules Apply The Realized Loss Is
Mar 28, 2026
-
The Secondary Market Is Best Defined As The Market
Mar 28, 2026