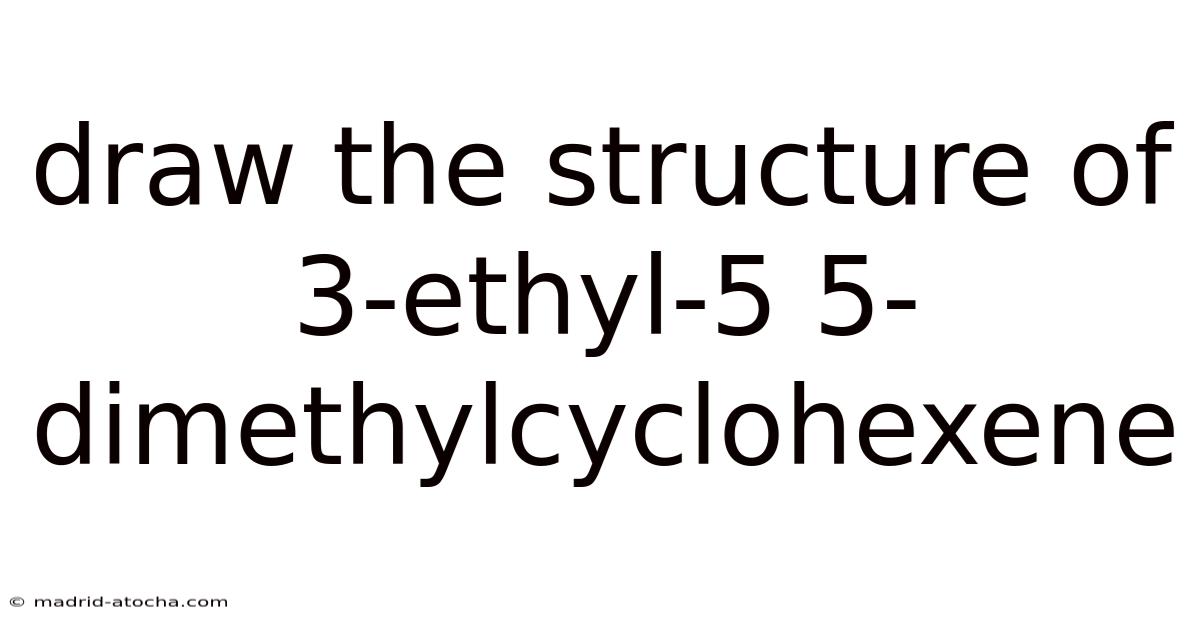
How to Draw the Structure of 3-Ethyl-5,5-Dimethylcyclohexene: A Complete Step-by-Step Guide
Understanding how to draw organic compound structures is a fundamental skill in chemistry, and 3-ethyl-5,5-dimethylcyclohexene is an excellent example to practice this skill. This compound demonstrates several important concepts in organic chemistry, including cycloalkenes, substituent positioning, and IUPAC naming conventions. In this thorough look, we will explore everything you need to know about drawing the structure of 3-ethyl-5,5-dimethylcyclohexene correctly.
Understanding the Compound Name
Before we begin drawing, it is essential to understand what each part of the name "3-ethyl-5,5-dimethylcyclohexene" tells us about the structure. The IUPAC naming system provides specific information about the molecular framework and all substituents attached to it.
The name can be broken down into three main components:
- Cyclohexene: This indicates a six-membered carbon ring (cyclohexane) with one double bond. The "-ene" suffix tells us there is one carbon-carbon double bond in the ring.
- 3-ethyl: An ethyl group (-CH₂CH₃) is attached to carbon number 3 of the ring.
- 5,5-dimethyl: Two methyl groups (-CH₃) are attached to carbon number 5 of the ring. The "5,5-" notation indicates that both methyl groups are on the same carbon atom.
Numbering System in Cyclohexene
In cyclohexene, the numbering system follows specific rules established by IUPAC. Plus, the double bond receives priority in numbering, which means carbon atoms 1 and 2 are the ones connected by the double bond. This creates the fundamental six-membered ring structure with one double bond between C1 and C2 Took long enough..
The numbering proceeds around the ring as follows:
- Carbon 1: One end of the double bond
- Carbon 2: The other end of the double bond
- Carbon 3: Located two positions away from C2
- Carbon 4: Opposite the double bond region
- Carbon 5: Three positions from C2
- Carbon 6: Returns to complete the ring
This systematic numbering ensures that substituents are located at the lowest possible numbers, which is why the ethyl group is at position 3 rather than position 5 if you were to count in the other direction.
Step-by-Step Drawing Guide
Step 1: Draw the Basic Cyclohexene Ring
Begin by drawing a hexagon to represent the six-membered carbon ring. Worth adding: remember that one side of the hexagon represents the double bond. You can draw this as either two parallel lines on one side of the hexagon or as a standard hexagon with a double bond indicator on one edge Not complicated — just consistent..
The cyclohexene ring consists of six carbon atoms, with each carbon having four bonds. The double bond occupies two of the bonding positions on adjacent carbons, meaning these two carbons have only one hydrogen each (or one substituent) attached to them, while the other four carbons have two hydrogens each (or one hydrogen and one substituent) Surprisingly effective..
Step 2: Locate and Mark Carbon Positions
Label the six vertices of your hexagon from 1 to 6, starting from one end of the double bond. Place the numbers lightly in pencil or mentally note their positions:
- C1 and C2: The double bond carbons (adjacent vertices)
- C3: Two carbons away from C2 in one direction
- C4: The carbon opposite the double bond
- C5: Three carbons away from C2
- C6: The final carbon completing the ring
Step 3: Add the Ethyl Group at Position 3
At carbon 3, you need to attach an ethyl group. Practically speaking, an ethyl group consists of two carbon atoms: one directly attached to the ring (the -CH₂- part) and one methyl carbon at the end (-CH₃). Draw a branch extending from C3 going outward from the ring, then add another carbon at the end of this branch Not complicated — just consistent..
The ethyl group structure is -CH₂-CH₃, so you will be adding two additional carbons to your structure at position 3. This means C3 now has three carbon bonds: one to C2, one to C4, and one to the first carbon of the ethyl group And that's really what it comes down to. That's the whole idea..
Step 4: Add the Two Methyl Groups at Position 5
At carbon 5, you need to attach two methyl groups. Consider this: since the name specifies "5,5-dimethyl," both methyl groups connect to the same carbon atom (C5). Each methyl group is simply -CH₃ The details matter here..
Draw two separate branches extending from C5, each ending with a -CH₃ group. These branches can point in different directions to show that they are separate substituents, though they both originate from the same carbon atom That alone is useful..
Step 5: Complete the Hydrogen Atoms
Finally, you need to add hydrogen atoms to complete the bonding. Each carbon in an organic compound must have four bonds. Count the bonds on each carbon and add hydrogen atoms where needed:
- C1 (double bond carbon): Already has one bond to C2 (double) and one bond to C6. Needs one more bond (one hydrogen).
- C2 (double bond carbon): Already has one bond to C1 (double) and one bond to C3. Needs one more bond (one hydrogen).
- C3: Bonds to C2, C4, and the ethyl group. Needs one hydrogen.
- C4: Bonds to C3 and C5. Needs two hydrogens.
- C5: Bonds to C4, C6, and two methyl groups. Needs one hydrogen.
- C6: Bonds to C1 and C5. Needs two hydrogens.
The Complete Structure Description
The final structure of 3-ethyl-5,5-dimethylcyclohexene can be described as a six-membered ring with one double bond between carbons 1 and 2. At carbon 3, an ethyl group (-CH₂CH₃) extends outward from the ring. At carbon 5, two methyl groups (-CH₃ each) are attached to the same carbon atom.
The molecular formula for this compound is C₁₀H₁₈. Now, you can verify this by counting: 6 carbons in the ring + 2 carbons in the ethyl group + 2 carbons in the two methyl groups = 10 carbons total. For hydrogens: the ring carbons have varying numbers of hydrogens depending on their substitution level, and adding up all the hydrogen atoms (including those on the substituents) gives you 18 Worth knowing..
Visual Representation
While I cannot draw images directly, here is a text-based representation showing the connectivity:
CH₃
|
CH₃-C
| \
CH₂ CH₂CH₃
| |
C=C C
/ |
CH₂ CH₂
| |
CH₂ CH
\ /
C
This representation shows the connectivity pattern, though in practice, you would draw this as a proper hexagonal ring structure with the double bond on one side and the substituents extending from their respective positions.
Common Mistakes to Avoid
When drawing 3-ethyl-5,5-dimethylcyclohexene, students often make several common errors:
-
Wrong position of the double bond: Remember that in cyclohexene, the double bond defines carbons 1 and 2. Always start your numbering from the double bond Easy to understand, harder to ignore..
-
Placing substituents on wrong carbons: The ethyl group goes specifically at position 3, and the two methyl groups go specifically at position 5. Double-check your numbering.
-
Forgetting that 5,5-dimethyl means two methyl groups on the same carbon: The "5,5-" notation indicates both methyl groups attach to carbon 5, not to different carbons numbered 5.
-
Incomplete hydrogen counting: Every carbon must have exactly four bonds. Always count and verify the final structure.
-
Drawing a cyclohexane instead of cyclohexene: The "-ene" suffix is crucial—it indicates the presence of a double bond.
Why This Structure Matters
Understanding how to draw 3-ethyl-5,5-dimethylcyclohexene builds important skills that apply to many other organic compounds. The principles used here—cycloalkene rings, substituent attachment, and systematic numbering—appear repeatedly in organic chemistry No workaround needed..
This particular compound also has practical relevance in organic synthesis and can serve as an intermediate in producing various chemical products. The ability to visualize and draw such structures is essential for understanding reaction mechanisms and predicting how molecules will behave in different chemical reactions Still holds up..
Frequently Asked Questions
What is the molecular formula of 3-ethyl-5,5-dimethylcyclohexene?
The molecular formula is C₁₀H₁₈. This consists of 10 carbon atoms and 18 hydrogen atoms Simple, but easy to overlook..
How many rings does this compound have?
This compound has one six-membered ring, as indicated by the "cyclohexene" part of the name.
Where is the double bond located?
The double bond is between carbon 1 and carbon 2 of the six-membered ring. This is standard for cyclohexene nomenclature Turns out it matters..
What does "5,5-dimethyl" mean?
The "5,5-dimethyl" notation indicates that two methyl groups are attached to the same carbon atom at position 5. This is called a gem-dimethyl substitution (from the Latin "gemellus" meaning twin) Small thing, real impact..
How do I know which way to number the ring?
In cyclohexene, numbering starts from the double bond to give it the lowest possible numbers. The substituents are then numbered to give the lowest set of locants.
Can the ring be drawn flat or does it have a specific 3D shape?
In reality, cyclohexene adopts a puckered conformation to relieve ring strain. On the flip side, for basic structural drawing purposes, a flat hexagonal representation is acceptable and commonly used Worth keeping that in mind..
Conclusion
Drawing the structure of 3-ethyl-5,5-dimethylcyclohexene is a straightforward process once you understand the naming system and follow a systematic approach. Remember these key points: start with a six-membered ring containing one double bond, number from the double bond, attach an ethyl group at position 3, and attach two methyl groups at position 5. Finally, complete all carbon bonding requirements with appropriate hydrogen atoms Simple as that..
This changes depending on context. Keep that in mind Small thing, real impact..
This skill forms the foundation for understanding more complex organic molecules and their reactions. Practice drawing this structure several times until it becomes automatic, and you will find that similar compounds become much easier to handle. The systematic approach to breaking down compound names and reconstructing molecular structures is invaluable throughout your study of organic chemistry Small thing, real impact..