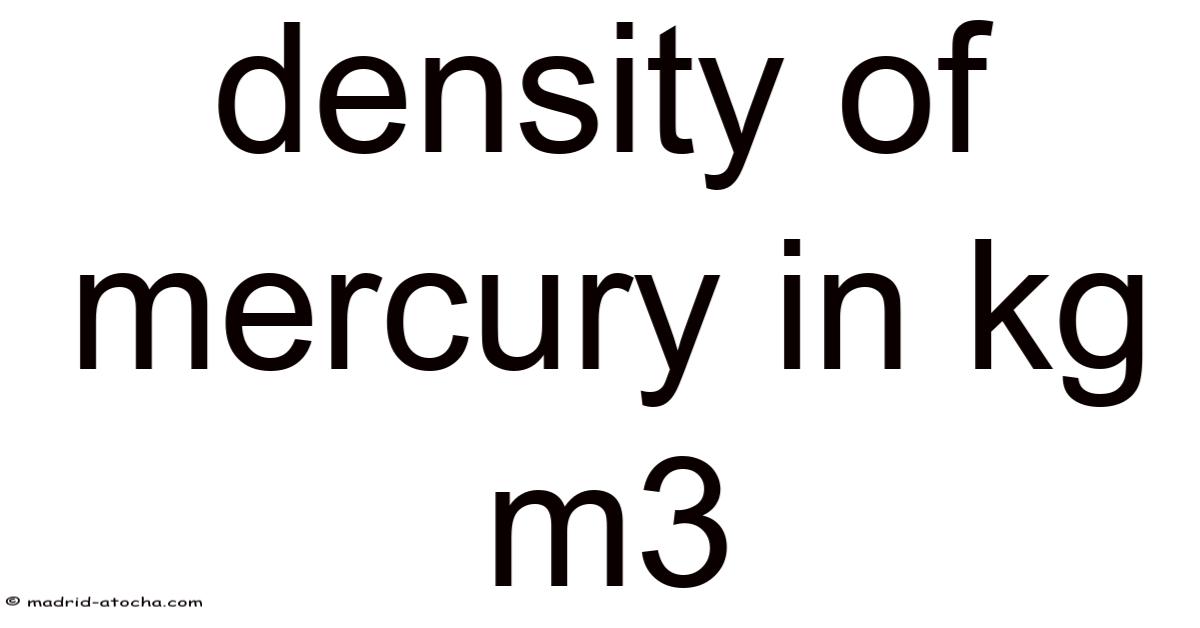The density of mercury in kg/m³ is one of those fundamental scientific constants that feels almost mythical in its significance. Even so, this extraordinary property isn’t just a trivia fact; it’s the cornerstone of its historical and modern applications, from thermometers to industrial processes. Day to day, at 13,534 kg/m³ at room temperature and standard atmospheric pressure, mercury is famously dense—over thirteen times denser than water. Understanding this number means understanding the very nature of this unique liquid metal, its behavior, and why it has captivated scientists and engineers for centuries Most people skip this — try not to..
The Scientific Explanation: Why So Dense?
To grasp why mercury is so dense, we must look at its atomic structure and the forces holding it together. But ” With an atomic number of 80, it sits in the transition metals on the periodic table. Mercury’s symbol, Hg, comes from its Greek name hydrargyrum, meaning “liquid silver.That said, unlike its metallic cousins like iron or gold, mercury’s electrons orbit the nucleus at incredibly high speeds due to relativistic effects. This “inward spiral” of electrons causes the outer electron shell to contract, weakening the metallic bonds between individual mercury atoms Worth keeping that in mind. Surprisingly effective..
The result? A metal with a very loose, easily disrupted crystal lattice. When mercury melts at −38.83 °C, it doesn’t form a tight, orderly solid structure. Instead, its atoms remain in a disordered, “liquid” arrangement even in the solid state (it’s one of the few metals that is liquid at room temperature). This disordered packing, combined with the substantial mass of each mercury atom (200.59 atomic mass units), means you can pack a tremendous amount of mass into a very small volume. That packed mass per unit volume is precisely its density: 13,534 kilograms per cubic meter.
Comparing Densities: Mercury in Context
Putting 13,534 kg/m³ into perspective helps cement its uniqueness. Here’s how mercury compares to other common substances:
- Water: 1,000 kg/m³ (Mercury is ~13.5 times denser)
- Iron: 7,870 kg/m³ (Mercury is denser than solid iron!)
- Lead: 11,340 kg/m³ (Mercury surpasses even lead)
- Gold: 19,320 kg/m³ (Denser, but mercury is no lightweight)
- Aluminum: 2,700 kg/m³
- Seawater: ~1,025 kg/m³
This extreme density is why a small flask of mercury feels surprisingly heavy and why it’s used in applications where weight and compactness are critical. In real terms, a one-liter bottle of mercury would weigh over 13. 5 kilograms—a palpable heft that demonstrates the concept of density in a tangible way Worth keeping that in mind..
Honestly, this part trips people up more than it should.
Measuring the Density of Mercury
The standard value of 13,534 kg/m³ is determined under specific standard conditions: 0 °C and 1 atm pressure. Consider this: this thermal expansion coefficient is well-documented and predictable, which is precisely why mercury was the fluid of choice for thermometers for so long. Even so, like all materials, the density of mercury changes slightly with temperature. Consider this: as temperature rises, mercury expands, and its density decreases. The predictable, linear expansion of mercury with heat allowed for accurate temperature measurement against a calibrated scale.
In a laboratory, density is calculated using the simple formula:
Density (ρ) = Mass (m) / Volume (V)
For mercury, you would weigh a precisely measured volume of the liquid in a flask of known volume (like a volumetric flask) and perform the division. Modern instruments like pycnometers or digital density meters can provide extremely precise measurements, accounting for temperature and pressure to give values that align with the accepted standard.
The Role of Density in Mercury’s Applications
The high density of mercury in kg/m³ is not a mere academic detail; it is the direct reason for its historical and niche modern uses Simple, but easy to overlook..
1. Barometers and Manometers: This is the most famous application. Atmospheric pressure can support a column of liquid. Because mercury is so dense, a column of mercury only needs to be about 760 mm (29.92 inches) high to balance standard atmospheric pressure. If water were used, the barometer would need to be over 10 meters tall. The specific gravity of mercury (its density relative to water) makes practical, compact pressure measurement possible.
2. Electrical Switches and Relays: In a mercury switch, a small amount of mercury rolls inside a sealed glass or metal bulb. When the switch is tilted, the mercury flows to make contact with two electrodes, closing the circuit. Its high density ensures that the mercury forms a clean, single, reliable bead that moves decisively, providing a positive on/off action without bouncing or arcing.
3. Industrial and Scientific Processes: Mercury’s density and liquid state at room temperature make it useful in diffusion pumps for creating high vacuums, in some types of batteries, and as a catalyst in chemical reactions. Its weight is also exploited in seismic dampers for buildings and in flywheel energy storage systems.
4. Gold and Silver Mining (Historically): The dense mercury forms an amalgam with gold and silver, binding to the precious metal particles. The amalgam is then separated from the less dense ore and heated to vaporize the mercury, leaving the pure metal behind. This process, called amalgamation, relies entirely on the density difference Surprisingly effective..
The Flip Side: Hazards of a Dense Liquid
The very properties that make mercury useful also make it dangerous. That's why, handling mercury requires stringent safety protocols, including ventilation, gloves, and specialized spill kits. That said, ** The phrase “mad as a hatter” originates from mercury poisoning suffered by 18th and 19th-century hat makers who used mercury nitrate to treat felt. Its high density means that spills are particularly problematic. Mercury forms tiny, rolling droplets that can seep into cracks, embed in fabrics, and evaporate slowly over time. The heavy, dense liquid is not easily absorbed through the skin in small amounts, but inhalation of vapor is the primary exposure route. **Mercury vapor is a potent neurotoxin.Its use is now heavily regulated or banned in many consumer applications due to these severe health and environmental risks Still holds up..
Frequently Asked Questions (FAQ)
Q: Is the density of mercury always 13,534 kg/m³? A: No, that is the standard density at 0 °C and 1 atm. It changes with temperature and pressure. For most practical calculations at room temperature (20–25 °C), a value of approximately 13,545 kg/m³ is often used.
Q: Why is mercury liquid at room temperature when other metals are solid? A: Due to relativistic effects on its inner electrons, mercury has very weak metallic bonds between its atoms, preventing it from forming a solid crystalline structure at standard temperatures It's one of those things that adds up..
Q: Can anything float on mercury? A: Yes, many common metals and materials are less dense than mercury and will float on its surface if placed gently. Iron, lead, silver, and even many types of stone will float because their densities are lower than 13,534 kg/m³ Worth knowing..
Q: Is mercury still used in thermometers? A: Its use in medical and consumer thermometers has been largely phased out in favor of safer alternatives like alcohol or digital sensors due to mercury’s toxicity. On the flip side, it may still be found in some laboratory or industrial thermometers The details matter here..
Q: How does the density of mercury compare to the densest element? A: The densest naturally occurring element is osmium, with a density of about 22,590 kg/m³—significantly denser than mercury. Even so, osmium is a solid, brittle
Q: How does the density of mercury compare to the densest element?
A: The densest naturally occurring element is osmium, with a density of about 22,590 kg/m³—significantly denser than mercury. On the flip side, osmium is a solid, brittle metal at room temperature. Mercury’s unique combination of high density and liquid state at ambient conditions makes it irreplaceable in niche applications like precision instruments and certain chemical processes, despite its dangers.
Q: What are the environmental impacts of mercury contamination?
A: Mercury released into the environment can convert into methylmercury, a highly toxic form that accumulates in fish and seafood. This bioaccumulation poses risks to wildlife and human health, particularly to developing fetuses and children. International treaties like the Minamata Convention aim to reduce mercury emissions and regulate its use in industries like mining and manufacturing Nothing fancy..
Conclusion
Mercury’s extraordinary density—13.5 times that of water—makes it a standout element in both industrial applications and scientific curiosity. From its role in gold extraction to its historical use in thermometers and pigments, the liquid metal has shaped human progress. Yet its toxicity and environmental persistence serve as stark reminders of the price of convenience. As we advance toward safer alternatives, mercury’s legacy underscores the importance of balancing utility with responsibility. Its story is not just one of density, but of duality: a element of remarkable strength and equally profound vulnerability.