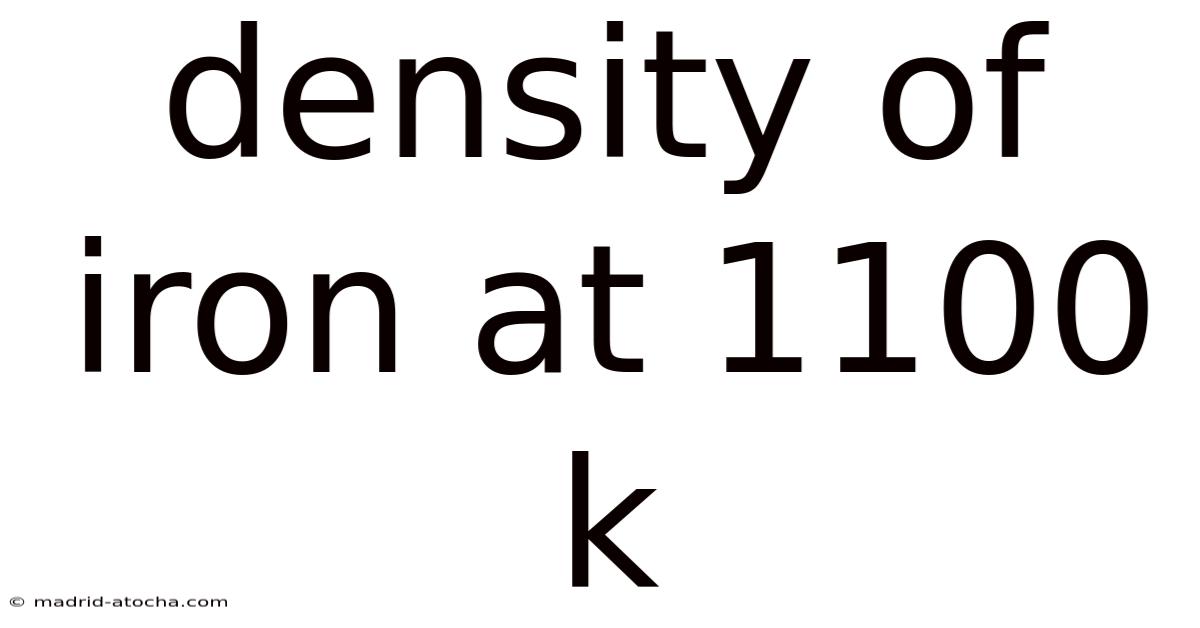Density of Iron at 1100 K: Understanding Thermal Effects on Material Properties
The density of iron at 1100 K is a critical parameter in materials science and engineering, particularly when studying how temperature influences the physical characteristics of metals. Which means for iron, a widely used metal in construction, manufacturing, and technology, its density at specific temperatures like 1100 K (1,832°F or 1,100°C) becomes especially relevant in high-temperature applications. At this elevated temperature, iron undergoes significant thermal expansion, altering its density and affecting its structural integrity, thermal conductivity, and mechanical performance. Density, defined as mass per unit volume, is a fundamental property that determines how substances interact with their environment. Understanding the density of iron at 1100 K requires examining the interplay between atomic vibrations, phase changes, and material behavior under stress Simple, but easy to overlook..
Factors Influencing the Density of Iron at 1100 K
The density of iron at 1100 K is not a static value but is influenced by several interrelated factors. Day to day, as iron is heated to 1100 K, its atoms gain kinetic energy, causing them to vibrate more vigorously. This increased atomic motion leads to thermal expansion, where the metal’s volume increases while its mass remains constant. Because of that, first, temperature itself plays a dominant role. Since density is inversely proportional to volume, this expansion results in a measurable decrease in density compared to its value at lower temperatures.
Second, the phase of iron at 1100 K is crucial. Iron exists in different crystalline structures, or phases, depending on temperature and pressure. At 1100 K, iron is in the austenite phase, a face-centered cubic (FCC) structure. That's why this phase is less dense than the body-centered cubic (BCC) ferrite phase found at lower temperatures. The transition between these phases affects how atoms are packed, directly impacting density. The FCC structure of austenite allows for more atomic spacing compared to the tightly packed BCC arrangement, contributing to a lower density at 1100 K That's the whole idea..
Third, impurities or alloying elements can alter the density. And pure iron has a specific density, but when combined with other metals or non-metals, the atomic arrangement and bonding change. Plus, for example, adding carbon to form steel modifies the crystal lattice, potentially increasing or decreasing density depending on the alloy composition. At 1100 K, these variations become more pronounced due to the heightened atomic activity.
Measuring the Density of Iron at 1100 K
Accurately determining the density of iron at 1100 K requires specialized techniques due to the extreme conditions involved. One common method is pycnometry, where a sample is weighed in a vacuum to eliminate air buoyancy effects. The sample is then submerged in a liquid medium at the desired temperature, and its volume is calculated based on displacement. Still, maintaining 1100 K in a controlled environment poses challenges, as most pycnometers operate at ambient temperatures Turns out it matters..
Another approach involves theoretical calculations using thermodynamic models. Scientists employ equations that account for thermal expansion coefficients and phase transformations to estimate density changes. These models rely on empirical data from experiments conducted at similar temperatures. On the flip side, for instance, the linear thermal expansion coefficient of iron is approximately 12. 1 × 10⁻⁶ per Kelvin. Using this value, the volume increase at 1100 K can be calculated, allowing for a density adjustment from the standard room-temperature value of about 7.87 g/cm³ Still holds up..
Practical applications also influence measurement methods. On the flip side, in industrial settings, where real-time data is critical, non-destructive testing techniques like X-ray diffraction (XRD) or ultrasonic testing may be used to infer density changes indirectly. These methods analyze structural properties that correlate with density, providing valuable insights without requiring direct volume measurements And that's really what it comes down to..
Scientific Explanation: Atomic Behavior at 1100 K
At the
Understanding the density of iron at 1100 K reveals much about its atomic behavior and material properties under extreme conditions. This structural shift increases atomic spacing, which typically correlates with a reduction in density despite the higher kinetic energy of the atoms. As temperature rises, iron transitions from the ferrite phase in cooler environments to austenite, which adopts a more open, face-centered cubic arrangement. This phenomenon underscores how macroscopic density can be intertwined with microscopic structural changes.
To build on this, the presence of alloying elements plays a central role in modifying density. So naturally, even slight additions of carbon or other metals can disrupt the iron lattice, either stiffening or softening it depending on the balance of forces at play. At 1100 K, these subtle adjustments become more noticeable, emphasizing the importance of precise composition in industrial materials And it works..
Measuring such subtle variations demands advanced instrumentation, blending experimental rigor with theoretical modeling. By combining empirical data with computational predictions, researchers can map density changes with precision, ensuring accurate characterization of iron across its thermal landscape Worth knowing..
All in all, the density of iron at 1100 K is a delicate balance of temperature, pressure, and composition, offering valuable insights into material science. This knowledge not only enhances our understanding of phase transitions but also guides the development of high-performance alloys That's the part that actually makes a difference..
Conclusion: The interplay of temperature, pressure, and alloy composition shapes iron’s density, highlighting the necessity of both experimental and theoretical approaches in material analysis.