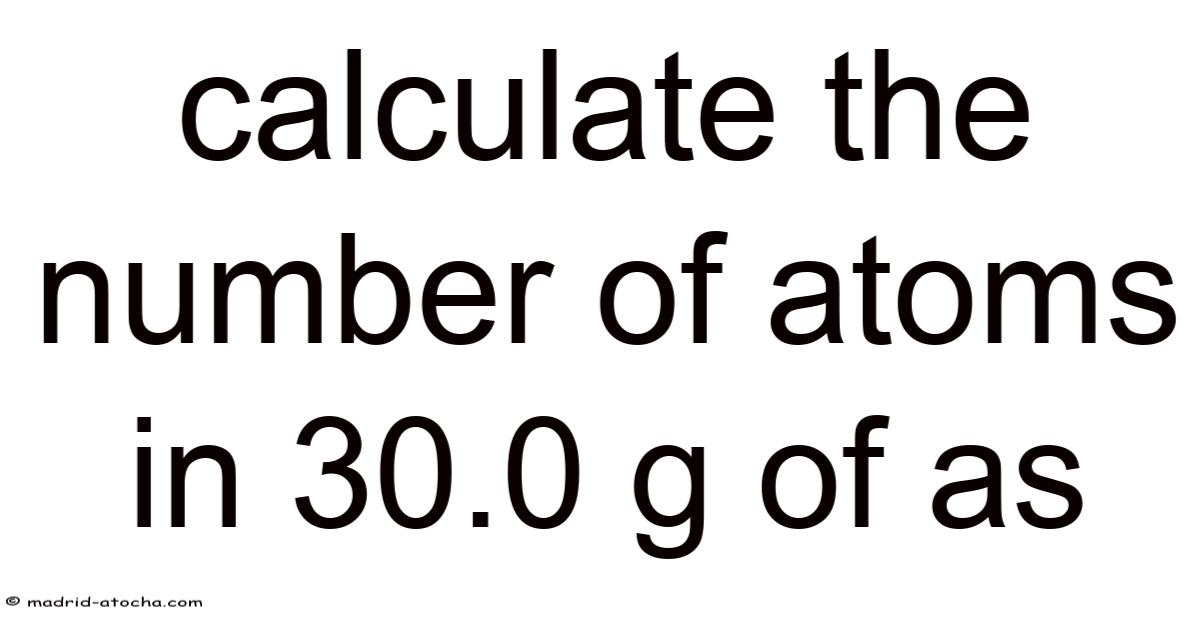Calculate the Number of Atoms in 30.0 g of Arsenic
Arsenic (As) is a chemical element that's part of the periodic table, and it's found in various compounds and minerals. Understanding the atomic structure of elements like arsenic is fundamental in chemistry, not just for academic purposes but also for practical applications in fields such as medicine, electronics, and environmental science. One common task in chemistry is to calculate the number of atoms in a given mass of an element. In this article, we will break down how to calculate the number of atoms in 30.0 grams of arsenic, providing a step-by-step guide to ensure clarity and understanding And that's really what it comes down to. And it works..
Introduction
The number of atoms in a given mass of an element can be calculated using the concept of moles. A mole is a fundamental unit in chemistry that represents a specific quantity of a substance. That's why one mole of any element contains Avogadro's number of atoms, which is approximately 6. On top of that, 022 x 10^23 atoms. This number is so large that it allows chemists to count atoms in a practical manner. By using the molar mass of an element, which is the mass of one mole of that element, we can determine the number of atoms in a given mass.
People argue about this. Here's where I land on it The details matter here..
Molar Mass of Arsenic
The first step in calculating the number of atoms in 30.Which means 0 grams of arsenic is to determine its molar mass. The molar mass of an element is numerically equal to its atomic weight, which can be found on the periodic table. For arsenic, the atomic weight is approximately 74.92 grams per mole. That's why this means that one mole of arsenic atoms weighs 74. 92 grams.
You'll probably want to bookmark this section.
Converting Grams to Moles
To find out how many moles are in 30.0 grams of arsenic, we use the formula:
[ \text{Number of moles} = \frac{\text{Mass of the substance (g)}}{\text{Molar mass (g/mol)}} ]
Plugging in the values, we get:
[ \text{Number of moles of As} = \frac{30.0, \text{g}}{74.92, \text{g/mol}} \approx 0.
Calculating the Number of Atoms
Now that we know there are approximately 0.400 moles of arsenic in 30.0 grams, we can calculate the number of atoms.
[ \text{Number of atoms} = \text{Number of moles} \times \text{Avogadro's number} ]
[ \text{Number of atoms of As} = 0.400, \text{mol} \times 6.022 \times 10^{23}, \text{atoms/mol} ]
[ \text{Number of atoms of As} \approx 2.4088 \times 10^{23}, \text{atoms} ]
Conclusion
By following these steps, we have calculated that there are approximately 2.Day to day, 4088 x 10^23 atoms in 30. That's why 0 grams of arsenic. Consider this: this calculation is a fundamental exercise in chemistry that demonstrates the relationship between mass, moles, and the number of atoms. Understanding this concept is crucial for anyone studying chemistry, as it forms the basis for more complex calculations involving chemical reactions, stoichiometry, and the composition of substances Easy to understand, harder to ignore..
FAQ
Q: Why is Avogadro's number so large? A: Avogadro's number is large because atoms are incredibly small. To have a practical number of atoms for counting, we use a large number that corresponds to the amount of substance in a mole Worth keeping that in mind..
Q: How does molar mass relate to atomic weight? A: The molar mass of an element in grams per mole is numerically equal to its atomic weight, which is the average mass of an atom of that element.
Q: Can I use the same method to calculate the number of atoms in other elements? A: Yes, you can use the same method to calculate the number of atoms in any element by using its molar mass and Avogadro's number.
By mastering these concepts, you'll be well-equipped to understand and calculate the number of atoms in various substances, a key skill in the field of chemistry But it adds up..
Applications of theCalculation
The ability to convert mass to moles and then to atoms is not just a theoretical exercise; it has practical implications in numerous scientific and industrial fields. Take this case: in environmental science, knowing the exact number of arsenic atoms in a sample helps assess contamination levels in water or soil. Arsenic is a toxic element, and even trace amounts can pose health risks. By calculating the atomic count, scientists can determine safe exposure thresholds and design remediation strategies. Similarly, in the pharmaceutical industry, precise measurements are critical when synthesizing arsenic-based compounds for medical use, such as certain chemotherapy drugs. The same principle applies to materials science, where understanding atomic composition aids in developing new alloys or semiconductors Which is the point..
Broader Implications in Chemistry
This calculation exemplifies a core principle in chemistry: the interplay between the macroscopic and microscopic worlds. While we measure substances in grams or liters, chemical reactions occur at the atomic level. Mastery of mole conversions allows chemists to predict reaction outcomes, balance chemical equations, and analyze substance behavior under different conditions. To give you an idea, stoichiometry—the calculation of reactant and product quantities—relies on these foundational concepts. Without the ability to translate mass into atomic counts, fields like analytical chemistry, pharmacology, and engineering would lack the precision needed for innovation and safety.
Conclusion
The process of determining the number of atoms in a given mass of arsenic underscores a fundamental truth in chemistry: the connection between measurable quantities and the invisible atomic structure of matter. By leveraging molar mass and Avogadro’s number, we bridge the gap between the tangible (grams) and the infinitesimal (atoms), enabling advancements in science and technology. Whether in environmental monitoring, drug development, or industrial applications, this calculation serves as a cornerstone of quantitative analysis. It reminds us that even the smallest units of matter play a key role in shaping our understanding of the world. As chemistry continues to evolve, the principles demonstrated here will remain essential, highlighting the enduring relevance of atomic-scale thinking in solving real-world challenges.