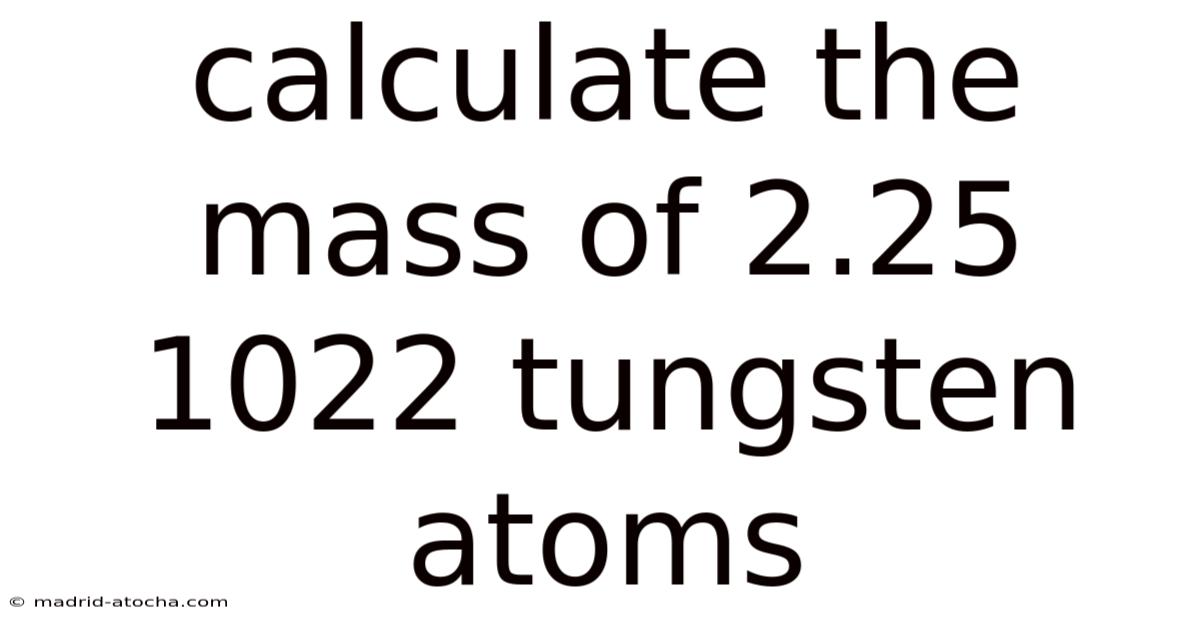How to Calculate the Mass of 2.25 × 10²² Tungsten Atoms
Calculating the mass of a specific number of atoms is one of the fundamental skills in chemistry that connects the microscopic world of individual atoms to the macroscopic world we can measure in the laboratory. Even so, when you need to determine the mass of 2. 25 × 10²² tungsten atoms, you'll need to apply Avogadro's number and the molar mass of tungsten in a systematic way. This calculation demonstrates how chemists bridge the gap between the countable world of atoms and the measurable world of grams and kilograms.
Understanding the Key Concepts
Before diving into the calculation itself, it's essential to understand the three key concepts that make this computation possible: atomic mass, molar mass, and Avogadro's number Simple, but easy to overlook..
Atomic mass refers to the mass of a single atom, typically expressed in atomic mass units (amu) or unified atomic mass units (u). For tungsten, the atomic mass is approximately 183.84 u, meaning a single tungsten atom has a mass of about 183.84 times the mass of one-twelfth of a carbon-12 atom And it works..
Molar mass is the mass of one mole of a substance. A mole is simply a counting unit, similar to a dozen, but instead of representing 12 items, it represents 6.022 × 10²³ items. The molar mass of tungsten is 183.84 grams per mole (g/mol), which numerically equals its atomic mass but expressed in grams rather than atomic mass units.
Avogadro's number (6.022 × 10²³) is the number of particles (atoms, molecules, or ions) in one mole of a substance. This constant is named after the Italian scientist Amedeo Avogadro and serves as the bridge between the atomic scale and the macroscopic scale we can measure.
Step-by-Step Calculation
Now let's work through the calculation to find the mass of 2.25 × 10²² tungsten atoms.
Step 1: Identify the Given Information
- Number of tungsten atoms: 2.25 × 10²² atoms
- Molar mass of tungsten (W): 183.84 g/mol
- Avogadro's number: 6.022 × 10²³ atoms/mol
Step 2: Convert Atoms to Moles
To find the mass, you first need to convert the number of atoms to moles using Avogadro's number:
$\text{Moles of W} = \frac{\text{Number of atoms}}{\text{Avogadro's number}}$
$\text{Moles of W} = \frac{2.25 \times 10^{22} \text{ atoms}}{6.022 \times 10^{23} \text{ atoms/mol}}$
$\text{Moles of W} = 0.03736 \text{ mol}$
Step 3: Calculate the Mass
Now multiply the moles of tungsten by its molar mass to obtain the mass:
$\text{Mass} = \text{Moles} \times \text{Molar mass}$
$\text{Mass} = 0.03736 \text{ mol} \times 183.84 \text{ g/mol}$
$\text{Mass} = 6.87 \text{ grams}$
Because of this, the mass of 2.25 × 10²² tungsten atoms is approximately 6.87 grams Still holds up..
Scientific Explanation: Why This Works
The calculation works because of the fundamental relationship between atoms, moles, and mass. This relationship exists due to how we define the mole and atomic mass units.
The mole was defined specifically so that the number of atomic mass units in one gram equals Avogadro's number. Also, this means that if you have a sample of an element with a mass equal to its atomic mass in grams, that sample will contain exactly Avogadro's number of atoms. Even so, for tungsten, this means 183. Practically speaking, 84 grams of tungsten contains exactly 6. 022 × 10²³ tungsten atoms.
This elegant relationship allows chemists to perform calculations by simply setting up proportions. Because of that, when you know the number of atoms you have, you can determine the number of moles by dividing by Avogadro's number. Then, multiplying by the molar mass gives you the actual mass in grams Small thing, real impact..
The calculation also demonstrates the dimensional analysis approach, where units are carried through the calculation to ensure the final answer has the correct units:
$\frac{2.25 \times 10^{22} \text{ atoms}}{1} \times \frac{1 \text{ mol}}{6.022 \times 10^{23} \text{ atoms}} \times \frac{183.84 \text{ g}}{1 \text{ mol}} = 6.
Practical Applications
Understanding how to calculate the mass of a specific number of atoms has numerous practical applications in chemistry and materials science.
In analytical chemistry, this calculation helps determine the purity of samples or the concentration of elements in compounds. When chemists perform analyses, they often need to know exactly how many atoms or molecules are present in their samples Most people skip this — try not to. Practical, not theoretical..
In materials science, calculating atom counts and masses is essential for understanding the composition of alloys, semiconductors, and nanomaterials. Tungsten, with its exceptionally high melting point (3422°C) and density (19.25 g/cm³), is widely used in applications requiring extreme heat resistance, such as light bulb filaments, welding electrodes, and radiation shielding.
The official docs gloss over this. That's a mistake.
In pharmaceutical chemistry, similar calculations determine the number of molecules in drug doses, helping researchers understand molecular interactions at the atomic level.
Frequently Asked Questions
Why do we use moles instead of counting atoms directly?
Counting atoms directly is impossible because atoms are far too small to count individually. The mole provides a practical way to work with manageable numbers while still representing a specific, fixed number of particles.
What if I need the answer in milligrams?
To convert grams to milligrams, multiply by 1000. So 6.87 grams equals 6870 milligrams.
Does the calculation change for different isotopes of tungsten?
Yes. The atomic mass of 183.Here's the thing — tungsten has several natural isotopes, including ¹⁸²W, ¹⁸³W, ¹⁸⁴W, and ¹⁸⁶W. 84 represents the weighted average of these isotopes. If you were working with a single isotope, you would use that isotope's specific atomic mass.
What is the significance of tungsten's high atomic mass?
Tungsten's high atomic mass (183.Practically speaking, 84) contributes to its exceptional density and makes it useful for applications requiring heavy, durable materials. Its high atomic mass also means that even relatively small numbers of atoms represent significant mass.
Conclusion
Calculating the mass of 2.Plus, 25 × 10²² tungsten atoms involves a straightforward two-step process: first converting atoms to moles using Avogadro's number, then converting moles to grams using the molar mass. The final answer of approximately 6.87 grams demonstrates how even a modest number of atoms represents a measurable mass That's the part that actually makes a difference..
This type of calculation is fundamental to chemistry because it illustrates the connection between the atomic scale and the macroscopic world we can observe and measure. Whether you're preparing for an exam, working in a laboratory, or simply curious about the behavior of matter, understanding how to perform these conversions is essential for anyone studying the chemical sciences Worth keeping that in mind. Worth knowing..
Some disagree here. Fair enough.
The beauty of this calculation lies in its simplicity and universal applicability—the same method works for any element, whether you're calculating the mass of hydrogen atoms, iron atoms, or any other element in the periodic table Still holds up..
The layered world of chemistry and materials science relies heavily on precise calculations, especially when dealing with elements like tungsten that play critical roles in technology and industry. And building on the foundation of understanding tungsten's properties, it becomes clear how these calculations extend beyond simple measurements. To give you an idea, in semiconductor manufacturing, the properties of materials such as tungsten are vital for ensuring durability and performance under intense conditions Less friction, more output..
When exploring similar problems, make sure to recognize the methods behind them. The process of determining the number of molecules in a pharmaceutical dose, for example, involves converting mass to moles and then to grams, which is a standard practice in both research and production. These steps ensure accuracy, highlighting the importance of precision in scientific work.
Worth adding, the principles applied here are not confined to one domain. Whether you're adjusting measurements for isotopic variations or converting units for lab-scale experiments, the core logic remains consistent. This adaptability underscores the value of mastering these calculations, as they form the backbone of countless real-world applications.
Boiling it down, the seamless integration of theory and practice in these calculations reinforces the significance of each step. From the atomic to the macroscopic scale, understanding these connections empowers scientists and engineers alike.
Pulling it all together, the ability to figure out these calculations not only enhances technical skills but also deepens our appreciation for the precision required in scientific endeavors. Embracing such knowledge bridges the gap between abstract concepts and tangible outcomes, making it an invaluable asset in modern science Small thing, real impact. Turns out it matters..