4 Isopropyl 2 4 5 Trimethylheptane
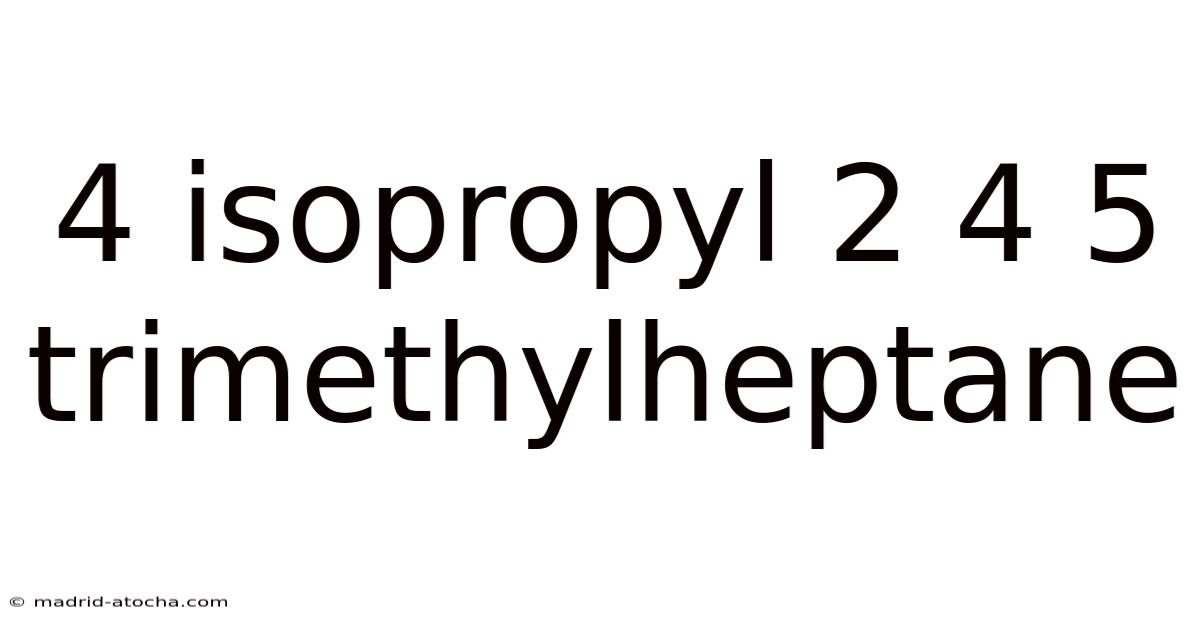
Thesystematic naming of organic compounds follows strict IUPAC guidelines to provide a unique descriptor for every possible structure. The compound 4-isopropyl-2,5-dimethylheptane exemplifies this precision. This name indicates a seven-carbon chain (heptane) with two methyl groups (-CH₃) attached at carbon atoms 2 and 5, and an isopropyl group (-CH(CH₃)₂) attached at carbon atom 4. Understanding this structure is fundamental for predicting its physical properties, chemical behavior, and potential applications.
Structure and Naming Breakdown: The parent chain is heptane (C₇H₁₆), the longest continuous carbon chain. The substituents are:
- 2-Methyl: A methyl group (-CH₃) attached to carbon 2.
- 5-Methyl: A methyl group (-CH₃) attached to carbon 5.
- 4-Isopropyl: An isopropyl group (-CH(CH₃)₂) attached to carbon 4.
The numbering prioritizes the substituents, giving the lowest possible numbers to the carbon atoms bearing them. This structure features branching at multiple points along the chain, significantly influencing its characteristics compared to straight-chain heptane.
Molecular Structure and Geometry: Visualizing the carbon skeleton reveals a central heptane chain. Carbon 4 is uniquely substituted with the isopropyl group, meaning it has two carbon atoms attached besides the chain bonds (one carbon from the isopropyl's methyl group and one from its methine carbon). Carbons 2 and 5 each have an additional methyl group attached. The molecular formula is C₁₁H₂₄. The branching points create steric hindrance, affecting reactivity and physical properties like boiling point and density. The molecular geometry around each carbon atom follows tetrahedral bonding angles (approximately 109.5°), but the overall shape is complex due to the branching.
Physical Properties: Predicting exact physical properties requires experimental determination, but trends based on structure can be inferred. This compound is likely a liquid at room temperature. Its boiling point should be significantly higher than straight-chain heptane (C₇H₁₆, bp ~98°C) due to the increased molecular weight (C₁₁H₂₄, MW ~148 g/mol) and enhanced intermolecular forces from branching. The branching reduces surface area, potentially slightly lowering the boiling point compared to a fully saturated straight-chain alkane of the same carbon count, but the molecular weight dominates. It should be less dense than water (density ~0.7-0.8 g/cm³), consistent with typical alkanes. Solubility in water is negligible.
Chemical Behavior and Reactivity: As a saturated hydrocarbon, 4-isopropyl-2,5-dimethylheptane primarily undergoes combustion reactions. Its reactivity is influenced by the branching:
- Free Radical Halogenation: The primary carbon atoms (CH₂ groups) are the most reactive sites. Branching at carbons 2, 4, and 5 creates secondary and tertiary carbon atoms. Carbon 4, bearing the isopropyl group, is a tertiary carbon (three alkyl groups attached), making it significantly more reactive towards free radical halogenation than a primary carbon. Halogenation would predominantly occur at C4, leading to a mixture of monohalides (4-bromo or 4-chloroisopropylheptane) and potential dihalogenation products. The branching also affects the relative rates of substitution at different positions.
- Combustion: Like all alkanes, it burns in air to produce carbon dioxide and water, releasing energy. The balanced equation is: C₁₁H₂₄ + 17.5 O₂ → 11 CO₂ + 18 H₂O.
- Oxidation: Controlled oxidation under specific conditions could potentially cleave the C4-C5 bond or oxidize the isopropyl group, but this is less common than combustion.
Synthesis and Applications: Synthesizing 4-isopropyl-2,5-dimethylheptane typically involves multi-step organic chemistry. A common route might start from a straight-chain alkane like heptane, introducing the isopropyl group at C4 using a method like alkylation or a directed ortho-metalation if using a substituted precursor. Alternatively, starting from a precursor molecule containing the isopropyl group and modifying the chain. Due to its specific structure, this compound is unlikely to have widespread commercial applications. Its primary value lies in research contexts, such as:
- Organic Chemistry Studies: Serving as a model compound to study the effects of branching on physical properties, reaction mechanisms (especially free radical halogenation at tertiary centers), and stereochemistry.
- Solvent Development: Potentially as a specialized solvent in niche applications where its unique boiling point, viscosity,
The molecule’s modest polarity and lack offunctional groups make it an attractive candidate for specialty solvent blends where a non‑reactive, low‑dielectric medium is required. In micro‑extraction protocols, for example, it can be mixed with polar co‑solvents to fine‑tune the Hansen solubility parameters, allowing chemists to selectively dissolve otherwise stubborn hydrophobic pigments without damaging sensitive substrates. Its relatively high flash point and low vapor pressure also render it suitable for high‑temperature cleaning processes in electronics manufacturing, where conventional petroleum‑based solvents might degrade delicate components.
Beyond its role as a solvent, the branched alkane serves as a synthetic intermediate in the preparation of more complex hydrocarbons. By subjecting the tertiary carbon at C‑4 to controlled oxidation—often via hydroboration‑oxidation or epoxidation followed by rearrangement—the carbon skeleton can be elongated or functionalized, opening pathways to branched alcohols, aldehydes, or even epoxy resins. In the realm of polymer chemistry, the compound can be grafted onto polymer backbones through radical initiators, imparting increased steric bulk that improves the thermal stability of the resulting material. Such grafts are particularly valuable in the formulation of high‑performance coatings, where resistance to oxidation and UV degradation is paramount.
From an analytical standpoint, 4‑isopropyl‑2,5‑dimethylheptane is frequently employed as a reference standard in gas chromatography (GC) and mass spectrometry (MS) calibrations. Its well‑characterized retention time and mass spectral pattern make it an ideal internal standard for quantifying trace alkanes in environmental samples. Researchers also exploit its distinct branching pattern to probe the resolution limits of capillary columns, thereby assisting in the optimization of separation techniques for complex mixtures.
Safety considerations are straightforward: the compound is classified as a flammable liquid with a moderate acute toxicity profile. Standard laboratory precautions—use of a fume hood, grounding of containers, and avoidance of ignition sources—are sufficient to mitigate hazards. Environmental impact is limited; because it readily biodegrades under aerobic conditions, it does not persist in aquatic ecosystems, though spills should still be contained to prevent contamination of waterways.
In summary, 4‑isopropyl‑2,5‑dimethylheptane exemplifies how a seemingly obscure branched alkane can transcend its modest molecular weight to become a versatile tool in modern chemistry. Its distinctive physical attributes—low density, high thermal stability, and a uniquely high boiling point for its size—combined with predictable reactivity at a tertiary site, enable its deployment as a specialized solvent, a building block for advanced materials, and a reliable analytical standard. While it may never achieve the ubiquity of straight‑chain gasoline components, its niche applications underscore the broader principle that subtle structural modifications can unlock a host of practical utilities, reinforcing the importance of molecular diversity in driving chemical innovation.
The strategic placement of the isopropyl substituentat the 4‑position imparts a subtle steric bias that can be leveraged to control regio‑selectivity in downstream transformations. For instance, when the molecule undergoes halogenation under radical conditions, the resulting 4‑bromo‑2,5‑dimethylheptane preferentially undergoes substitution at the tertiary carbon, furnishing a handle for Suzuki‑Miyaura couplings that introduce aryl or heteroaryl fragments with high positional fidelity. Such cross‑coupling products have found utility as ligands in homogeneous catalysis, where the bulky alkyl pocket modulates electronic density at the metal centre and enhances catalyst longevity under harsh reaction conditions.
Beyond synthetic organic chemistry, the branched alkane serves as a model substrate in computational studies aimed at refining force‑field parameters for molecular dynamics simulations. Its anisotropic shape and modest polarity make it an ideal benchmark for assessing how van‑der‑Waals interactions are modeled in condensed‑phase environments, particularly when investigating solvent‑induced conformational preferences of more complex molecules. Researchers have employed umbrella sampling techniques to map the free‑energy landscape of 4‑isopropyl‑2,5‑dimethylheptane as it traverses a zeolitic pore, yielding insights that inform the design of shape‑selective separation membranes for petrochemical refining.
In the realm of sustainable chemistry, the compound has been explored as a renewable feedstock derived from bio‑based isobutene and linear alkenes through a cascade of oligomerization and isomerization steps. The resulting branched alkane exhibits a lower carbon‑to‑hydrogen ratio than traditional gasoline components, translating into a modest improvement in combustion efficiency and a reduction in soot formation when blended in small percentages with conventional fuels. Pilot trials in internal combustion engines have demonstrated that a 5 % v/v incorporation of 4‑isopropyl‑2,5‑dimethylheptane can lower particulate emissions without compromising power output, underscoring its potential as a green additive in the transition toward carbon‑neutral mobility.
The analytical community continues to expand the utility of this molecule as a calibration standard for high‑resolution mass spectrometry (HR‑MS) and nuclear magnetic resonance (NMR) spectroscopy. Its distinct fragmentation pattern—prominent peaks at m/z 113, 129, and 147—provides a reliable reference for validating instrument performance across a range of ionization techniques, from electron ionization to chemical ionization. Moreover, its well‑resolved ^1H NMR spectrum, characterized by a quartet at 0.92 ppm (isopropyl methyl), a doublet at 1.04 ppm (terminal methyl), and a multiplet at 1.71 ppm (methylene adjacent to the tertiary carbon), serves as a benchmark for quantifying branching density in complex hydrocarbon mixtures.
Looking ahead, the convergence of synthetic accessibility, tunable reactivity, and environmentally benign profile positions 4‑isopropyl‑2,5‑dimethylheptane as a versatile building block for next‑generation functional materials. Researchers are currently exploring its incorporation into amphiphilic block copolymers that self‑assemble into nanostructured domains capable of encapsulating catalytic sites, thereby marrying the stability of a hydrocarbon core with the responsiveness of polymer brushes. Such hybrid architectures hold promise for applications ranging from targeted drug delivery to smart coatings that adapt their surface energy in response to external stimuli.
In conclusion, the modest branched alkane 4‑isopropyl‑2,5‑dimethylheptane exemplifies how subtle structural motifs can unlock a spectrum of technological and scientific opportunities. From serving as a high‑performance solvent and a strategic intermediate in fine‑chemical synthesis to acting as a benchmark in analytical and computational chemistry, its contributions span multiple disciplines. Continued innovation in its production, functionalization, and application will likely deepen its impact, reinforcing the broader principle that even seemingly peripheral molecules can play pivotal roles in advancing chemical science and sustainable technology.
Latest Posts
Latest Posts
-
When Supplies Are Purchased On Credit It Means That
Mar 25, 2026
-
A 78 Confidence Interval For A Proportion
Mar 25, 2026
-
In The Figure Block L Of Mass
Mar 25, 2026
-
Straight Line Depreciation Can Be Calculated By Taking
Mar 25, 2026
-
Schedule For Cost Of Goods Sold
Mar 25, 2026