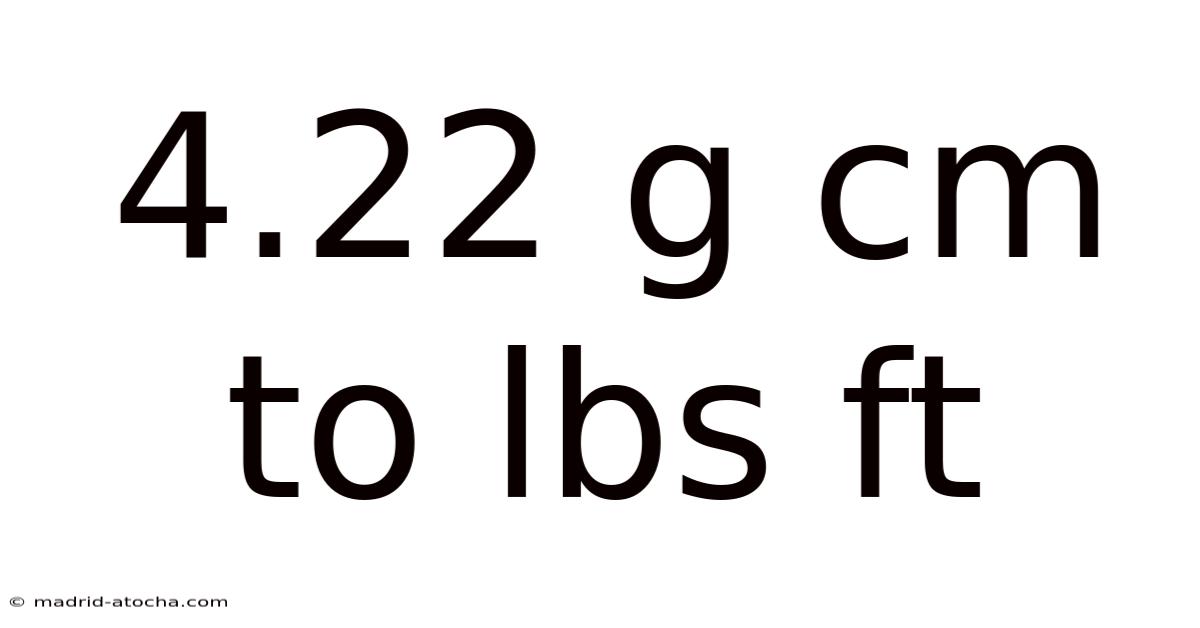Converting between metric and imperial units can be confusing, especially when dealing with density measurements like grams per cubic centimeter (g/cm³) and pounds per cubic foot (lbs/ft³). Even so, understanding how to convert 4. 22 g/cm³ to lbs/ft³ is essential in fields such as engineering, construction, and materials science. This article will guide you through the conversion process step by step, explain the science behind it, and provide practical examples to help you master this skill.
Understanding the Units
Before diving into the conversion, make sure to understand what these units represent. Grams per cubic centimeter (g/cm³) is a metric unit of density, commonly used in scientific contexts. But it measures mass per unit volume. On the flip side, on the other hand, pounds per cubic foot (lbs/ft³) is an imperial unit of density, often used in industries like construction and shipping. It also measures mass per unit volume but uses different base units.
Not the most exciting part, but easily the most useful.
The Conversion Process
To convert 4.22 g/cm³ to lbs/ft³, you need to use a conversion factor. The key is to convert grams to pounds and cubic centimeters to cubic feet.
- Convert grams to pounds: 1 gram is approximately 0.00220462 pounds.
- Convert cubic centimeters to cubic feet: 1 cubic centimeter is approximately 0.0000353147 cubic feet.
Now, let's apply these conversion factors to 4.22 g/cm³:
[ 4.22 , \text{g/cm}^3 \times 0.00220462 , \text{lbs/g} \times \frac{1}{0 The details matter here. And it works..
Breaking it down:
[ 4.22 \times 0.00220462 = 0.009304 , \text{lbs/cm}^3 ]
[ \frac{0.009304}{0.0000353147} = 263.6 , \text{lbs/ft}^3 ]
So, 4.22 g/cm³ is approximately 263.6 lbs/ft³ Worth keeping that in mind..
Why This Conversion Matters
Understanding how to convert between these units is crucial in many real-world applications. Here's one way to look at it: in construction, materials like concrete or steel are often specified in lbs/ft³, while scientific research might use g/cm³. Being able to switch between these units ensures accuracy in calculations and communication across different industries Practical, not theoretical..
Common Mistakes to Avoid
When performing conversions, it's easy to make mistakes. Here are some common pitfalls to watch out for:
- Forgetting to cube the conversion factor: When converting volume units, remember to cube the linear conversion factor. Here's one way to look at it: converting cm to ft requires cubing the factor because you're dealing with cubic units.
- Mixing up mass and volume conversions: Ensure you're converting both mass (grams to pounds) and volume (cubic centimeters to cubic feet) correctly.
- Rounding too early: Keep as many decimal places as possible during intermediate steps to avoid rounding errors in the final result.
Practical Examples
Let's look at a few practical examples where this conversion might be useful:
- Material Density: If you're working with a material that has a density of 4.22 g/cm³, knowing it's equivalent to 263.6 lbs/ft³ can help you compare it with other materials specified in imperial units.
- Shipping Calculations: When calculating the weight of a shipment, converting the density to lbs/ft³ can simplify the process if the shipping company uses imperial units.
- Engineering Design: In structural engineering, converting densities ensures that all calculations are consistent, regardless of the unit system used.
Frequently Asked Questions
Q: Why do we need to convert between metric and imperial units?
A: Different industries and countries use different unit systems. Converting between them ensures consistency and accuracy in global communication and calculations.
Q: Can I use an online converter for this?
A: Yes, online converters are convenient, but understanding the manual process helps you verify results and catch errors.
Q: What if I need to convert other density units?
A: The same principles apply. Identify the conversion factors for mass and volume, then apply them step by step Not complicated — just consistent..
Conclusion
Converting 4.22 g/cm³ to lbs/ft³ is a straightforward process once you understand the conversion factors and steps involved. Practically speaking, remember to double-check your calculations and be mindful of common mistakes to ensure accuracy. Consider this: by mastering this skill, you can confidently work with density measurements in both metric and imperial systems, enhancing your versatility in various professional fields. With practice, these conversions will become second nature, empowering you to tackle more complex problems with ease.
Beyond the Basics: Advanced Considerations
While the core conversion remains consistent, several nuances can arise depending on the context. Consider these advanced points:
- Temperature Dependence: Density is often temperature-dependent. The values provided (like 4.22 g/cm³) are typically at a specific temperature (often 20°C or 25°C). If your application involves significantly different temperatures, you'll need to account for thermal expansion or contraction, which will alter the density. This requires consulting material property tables or using more complex equations.
- Pressure Dependence (for Gases): For gases, density is highly sensitive to pressure. The conversion factors presented here are generally applicable at standard atmospheric pressure. If dealing with compressed or rarefied gases, pressure corrections are essential.
- Mixtures and Alloys: The density of a mixture or alloy isn't simply the weighted average of the component densities. Interactions between the components can lead to deviations. Specialized formulas or experimental measurements are needed for accurate density determination in these cases.
- Significant Figures: Always pay attention to significant figures. The number of significant figures in your final answer should be consistent with the least precise input value. Here's one way to look at it: if your initial density value (4.22 g/cm³) has only three significant figures, your final answer shouldn't be presented with five or six.
- Software Tools & Libraries: For repetitive or high-volume conversions, consider utilizing specialized software packages or programming libraries. These tools often incorporate temperature and pressure corrections and can handle complex mixtures, significantly reducing the risk of manual errors. Python libraries like
pintare particularly useful for unit handling and conversions.
Resources for Further Learning
- NIST (National Institute of Standards and Technology): Provides comprehensive data on physical properties, including density, for various materials. (https://www.nist.gov/)
- Engineering ToolBox: Offers a wide range of engineering data, including density values and conversion factors. (https://www.engineeringtoolbox.com/)
- Online Unit Conversion Calculators: While useful for quick checks, always understand the underlying principles. (Numerous options available – search for "metric to imperial unit converter")
- Textbooks on Physical Properties of Materials: Provide in-depth explanations of density and its behavior under different conditions.
Conclusion
Converting 4.Plus, by mastering this skill, you can confidently work with density measurements in both metric and imperial systems, enhancing your versatility in various professional fields. Remember to double-check your calculations, be mindful of common mistakes, and consider the advanced factors discussed – temperature, pressure, and mixture behavior – to ensure accuracy. On the flip side, the seemingly simple conversion belies a deeper understanding of physical properties and the potential complexities that can arise in real-world applications. This leads to 22 g/cm³ to lbs/ft³ is a straightforward process once you understand the conversion factors and steps involved. With practice, these conversions will become second nature, empowering you to tackle more complex problems with ease, and ultimately, contributing to more reliable and consistent results across diverse industries.
Common Pitfalls and How to Avoid Them
| Pitfall | Why It Happens | Quick Fix |
|---|---|---|
| Skipping the unit‑by‑unit breakdown | It’s tempting to multiply all the numbers in one go, but this can lead to mis‑placement of decimal points. | |
| Using the wrong temperature correction factor | Density tables often list values at standard laboratory conditions (20 °C, 1 atm). Confusing it with “lb force” can introduce a factor of g ≈ 32. | |
| Mixing up “lbm” and “lb” | In the US customary system, “lb” is a unit of mass (pound‑mass). Practically speaking, | |
| Ignoring significant figures | Reporting a result with too many digits gives a false impression of precision. On the flip side, | Write out each conversion step on paper or a spreadsheet. In practice, 174 ft/s². But applying a factor for a different temperature without checking the source can skew results. Here's the thing — |
Practical Example: Density of a Polymethyl Methacrylate (PMMA) Sample
- Given: ρ = 1.18 g/cm³ (at 25 °C, 1 atm).
- Convert:
- 1 g/cm³ = 62.42796 lbm/ft³.
- 1.18 g/cm³ × 62.42796 = 73.626 lbm/ft³.
- Temperature correction (if needed): Suppose you need the density at 70 °C, and the linear thermal expansion coefficient for PMMA is 70 × 10⁻⁶ /°C.
- ΔT = 70 °C – 25 °C = 45 °C.
- Relative volume change ≈ 70 × 10⁻⁶ × 45 ≈ 0.00315 (0.315 %).
- New density ≈ 73.626 / (1 + 0.00315) ≈ 73.35 lbm/ft³.
This illustrates how a seemingly negligible temperature change can affect the final figure by several tenths of a pound‑mass per cubic foot.
Integration into Engineering Workflows
- Design of Hydraulic Systems: Accurate fluid densities are vital for pump sizing and pressure drop calculations.
- Aerospace Component Analysis: Weight assessments rely on precise material densities; even a 1 % error can alter mission budgets.
- Environmental Monitoring: Density conversions are needed when reporting pollutant concentrations in different units for regulatory compliance.
By embedding a standardized conversion routine—ideally automated within your CAD or simulation software—you reduce the risk of human error and ensure consistency across projects.
Final Thoughts
While the arithmetic of converting 4.Because of that, 22 g/cm³ to pounds per cubic foot is elementary, the surrounding context—temperature, pressure, mixture composition, and significant figures—adds layers of nuance that professionals must work through. Mastery of these details transforms a simple unit conversion into a reliable foundation for engineering judgment, scientific reporting, and cross‑disciplinary collaboration.
In practice, adopt a habit of documenting the assumptions (e.Now, , reference temperature, pressure, and material purity) whenever you report density values. Also, g. take advantage of the solid libraries and databases mentioned earlier to keep your data current, and automate repetitive conversions whenever possible. With these strategies, you’ll convert not only units but also complex real‑world conditions into actionable, trustworthy numbers that drive successful outcomes across any field that relies on precise material characterization.