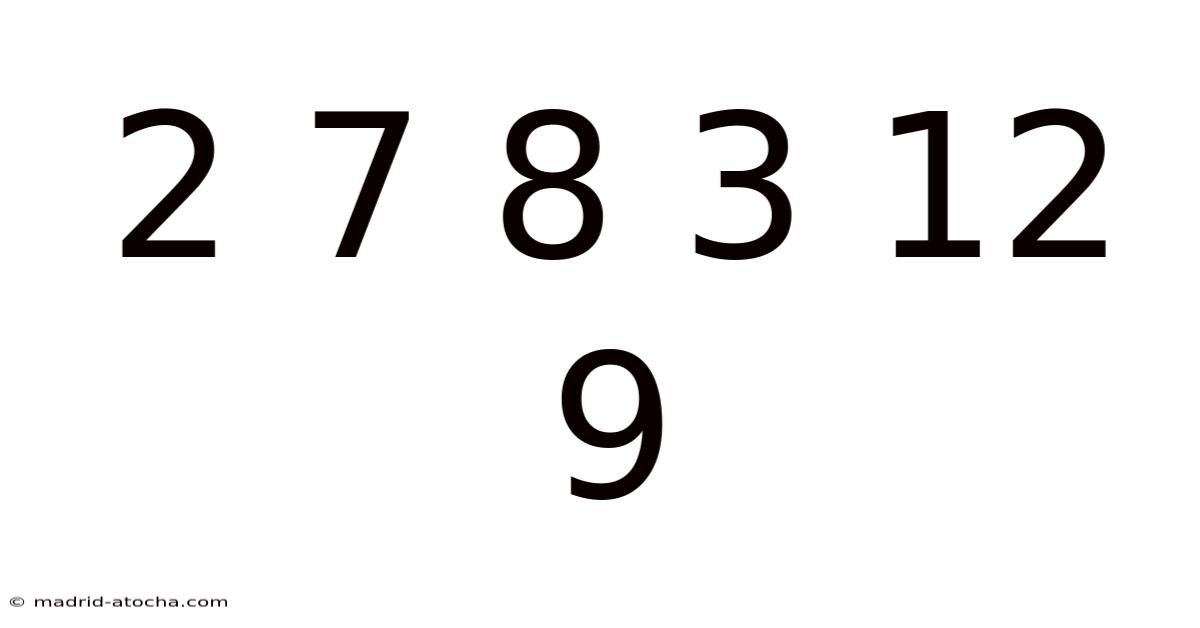2 7 8 3 12 9: Unlocking the Secrets of Six Key Elements and Their Everyday Impact
The sequence 2 7 8 3 12 9 may appear as a random string of digits, but within the realm of chemistry it represents the atomic numbers of six fundamental elements that shape our world. This article explores each number, reveals the element it denotes, and explains why these particular atoms are essential to life, industry, and technology. By the end, readers will not only understand the scientific significance of 2 7 8 3 12 9 but also discover practical ways to remember and apply this knowledge in education, DIY projects, and scientific literacy Not complicated — just consistent. That's the whole idea..
Not obvious, but once you see it — you'll see it everywhere.
The Numerical Code: Why These Digits MatterAt first glance, 2 7 8 3 12 9 looks like a simple counting exercise. In the periodic table, however, each integer corresponds to a unique element’s position based on the number of protons in its nucleus. Recognizing this connection transforms a bland list of numbers into a gateway for exploring:
- Helium (2) – the noble gas that keeps balloons afloat.
- Nitrogen (7) – the dominant component of the atmosphere we breathe.
- Oxygen (8) – the life‑supporting gas essential for respiration.
- Lithium (3) – a soft metal powering modern batteries.
- Magnesium (12) – the sturdy metal used in alloys and fireworks.
- Fluorine (9) – the highly reactive halogen found in toothpaste.
Understanding 2 7 8 3 12 9 therefore means grasping the basic building blocks of matter that influence everything from health to manufacturing.
The Elements Behind the Numbers
Helium – Atomic Number 2
Helium is a colorless, odorless noble gas that occupies the second slot on the periodic table. Day to day, its full valence shell makes it chemically inert, which is why it is widely used to fill party balloons, protect sensitive electronics, and cool superconducting magnets in MRI machines. Because it does not react with other substances, helium is also the preferred carrier gas in gas chromatography for laboratory analysis.
It sounds simple, but the gap is usually here.
Nitrogen – Atomic Number 7
Nitrogen makes up about 78 % of Earth’s atmosphere. This stability allows nitrogen to serve as a protective atmosphere in food packaging, electronics manufacturing, and even in the production of ammonia for fertilizers. Its diatomic form, N₂, is incredibly stable due to a strong triple bond between the two nitrogen atoms. In biology, nitrogen is a core component of amino acids and nucleic acids, making it indispensable for life.
Oxygen – Atomic Number 8
Oxygen’s atomic number of 8 places it in the same period as nitrogen but gives it a higher electronegativity. The molecule O₂ fuels cellular respiration, while ozone (O₃) forms a protective layer in the stratosphere that shields us from harmful ultraviolet radiation. Industrially, oxygen is crucial for steelmaking, wastewater treatment, and medical oxygen therapy.
Lithium – Atomic Number 3
Lithium is the lightest metal and the third element on the periodic table. Its low density and high electrochemical potential make it ideal for rechargeable batteries used in smartphones, electric vehicles, and portable electronics. Lithium compounds also serve as mood‑stabilizing agents in psychiatric treatment, highlighting the element’s diverse roles in both technology and health.
Magnesium – Atomic Number 12
Magnesium is a silvery-white alkaline earth metal that burns with a bright flame, a property exploited in flares and pyrotechnics. In the human body, magnesium is essential for over 300 enzymatic reactions, including those that regulate muscle and nerve function. Industrially, magnesium alloys are prized for their lightweight strength in aerospace and automotive components No workaround needed..
Fluorine – Atomic Number 9
Fluorine is the most electronegative element, and its highly reactive nature is harnessed in the formation of compounds such as Teflon, freon refrigerants, and dental fluorides. While elemental fluorine is too dangerous for direct use, its derivatives are integral to everyday products ranging from non‑stick cookware to toothpaste that helps prevent cavities.
Practical Applications in Daily Life
The elements tied to 2 7 8 3 12 9 are not confined to laboratory tables; they permeate our daily routines:
- Energy Storage – Lithium‑ion batteries power laptops, electric cars, and renewable energy storage systems.
- Healthcare – Helium cools MRI machines; magnesium treats arrhythmias; fluorine’s compounds protect teeth.
- Food Preservation – Nitrogen creates inert atmospheres
From the foundational role of nitrogen in maintaining our atmosphere to the vital functions of oxygen in sustaining life, each element contributes uniquely to the systems we rely on. Lithium’s lightness and reactivity make it indispensable in modern energy storage, while magnesium supports essential biological processes and enhances material strength. Fluorine’s extraordinary reactivity underlies up-to-date industrial and dental solutions, underscoring how the periodic table shapes our technological progress. Practically speaking, together, these elements illustrate the layered balance between stability and dynamism, resilience and transformation. Understanding their properties not only deepens our appreciation of chemistry but also inspires innovations that improve quality of life. In this way, the study of these elements bridges the scientific and the everyday, reminding us of the profound impact of atomic structure on our world Worth keeping that in mind..
Conclusion: The seamless integration of these key elements into everyday applications highlights their irreplaceable value. From powering our devices to safeguarding our health, recognizing their roles empowers us to harness science more effectively. This interconnectedness reinforces the importance of continued exploration and appreciation of the elements that underpin modern civilization Simple as that..
Oxygen – Atomic Number 8
Oxygen is arguably the most crucial element for life as we know it. Worth adding: it’s a fundamental component of water, the solvent of life, and is involved in cellular respiration, the process by which organisms convert food into energy. Its ability to readily form oxides is also utilized in steel production, creating stronger and more durable materials. Beyond that, oxygen’s properties are leveraged in combustion, fueling everything from industrial furnaces to rocket propulsion.
Quick note before moving on.
Sodium – Atomic Number 11
Sodium, a highly reactive alkali metal, plays a surprising role in food preservation. Which means when added to brine solutions, it inhibits the growth of bacteria, extending the shelf life of pickles and other preserved foods. Beyond the culinary world, sodium chloride (table salt) is a vital electrolyte for maintaining fluid balance in the human body and is essential for numerous industrial processes.
Potassium – Atomic Number 19
Potassium is another essential mineral, playing a critical role in nerve function, muscle contraction, and maintaining healthy blood pressure. It’s abundant in fruits and vegetables and is often supplemented to address deficiencies. Industrially, potassium compounds are used in fertilizers, boosting crop yields, and in the production of ceramics and glass.
Chlorine – Atomic Number 17
Chlorine’s potent disinfecting properties are utilized extensively in water treatment, ensuring safe drinking water supplies. It’s also a key ingredient in the production of PVC plastics, a versatile material used in construction, plumbing, and countless other applications. While elemental chlorine is toxic, its derivatives are essential for a wide range of products And that's really what it comes down to..
Argon – Atomic Number 18
Argon, a noble gas, is inert and non-reactive, making it invaluable as a shielding gas in welding, preventing oxidation and ensuring strong, clean welds. It’s also used in light bulbs to prolong filament life and in various analytical techniques.
Silicon – Atomic Number 14
Silicon, the second most abundant element in the Earth’s crust, is the backbone of the semiconductor industry. It’s the foundation of microchips and integrated circuits, the brains behind computers, smartphones, and countless other electronic devices. Beyond electronics, silicon is used in solar panels, capturing sunlight and converting it into electricity, and in construction materials like concrete No workaround needed..
Phosphorus – Atomic Number 15
Phosphorus is a vital component of DNA and RNA, the building blocks of life. Which means it’s also a key nutrient for plant growth, forming essential proteins and cell membranes. Industrially, phosphorus compounds are used in fertilizers, detergents, and flame retardants.
Conclusion: The elements identified – lithium, magnesium, fluorine, nitrogen, oxygen, sodium, potassium, chlorine, argon, silicon, and phosphorus – represent a remarkable spectrum of properties and applications. Their presence, often unseen and unacknowledged, is fundamental to the functioning of our planet and the advancement of human civilization. From the microscopic world of cellular processes to the macroscopic scale of industrial production and technological innovation, these elements demonstrate a profound and enduring influence. Their interconnectedness highlights a continuous cycle of transformation and utility, solidifying the periodic table’s position as a cornerstone of scientific understanding and a key to unlocking future possibilities.